Nature Catalysis, Published online: 23 September 2019; doi:10.1038/s41929-019-0346-z
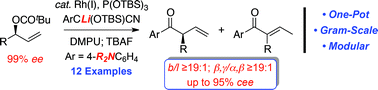
Asymmetric routes for the formation of tetrasubstituted allenes are scarce and limited in scope. Now, a catalytic asymmetric process is reported giving access to tetrasubstituted allenes from readily available propargylic alcohols, and a potential role for an achiral supporting ligand is postulated.ChusSpringsteen
Shared posts
Tetrasubstituted allenes via the palladium-catalysed kinetic resolution of propargylic alcohols using a supporting ligand
[ASAP] Homochiral Metal–Organic Frameworks for Enantioselective Separations in Liquid Chromatography
[ASAP] Transition-Metal-Free Allylic Borylation of 1,3-Dienes
[ASAP] Enol Acetates: Versatile Substrates for the Enantioselective Intermolecular Tsuji Allyation
[ASAP] Cu-Catalyzed Enantioselective Ring Opening of Cyclic Diaryliodoniums toward the Synthesis of Chiral Diarylmethanes
[ASAP] Enantioselective Copper-Catalyzed Defluoroalkylation Using Arylboronate-Activated Alkyl Grignard Reagents
[ASAP] Stereospecific Ring Contraction of Bromocycloheptenes through Dyotropic Rearrangements via Nonclassical Carbocation–Anion Pairs
Enantioselective Synthesis of Boryl Tetrahydroquinolines via Cu-Catalyzed Hydroboration
Single or Synergistic Kinetic Resolutions of Chiral Allylalanes: Two Complementary Routes for the Asymmetric Synthesis of Syn Homoallylamines
Chiral Pincer Carbodicarbene Ligands for Enantioselective Rhodium-Catalyzed Hydroarylation of Terminal and Internal 1,3-Dienes with Indoles
Frontispiece: Dimensioning the Term Carbenoid

A carbenoid species consists of a tetrasubstituted carbon atom bearing a leaving group X and a metal–carbon bond, from which extrusion of X triggers the elimination/transfer of the CR1R2 unit. This is the case of the largely known Simmons–Smith reaction (based on Zn) or the more recent gold-based carbenoid LAuCH2Cl described by Echavarren and co-workers. In the context of metal-induced carbene transfer from a diazo compound, a clear distinction must be made between the transient carbenoid and metal–carbene intermediates. For more information see the Concept article by A. Caballero and P. J. Pérez on page 14389 ff.
Computationally Guided Catalyst Design in the Type I Dynamic Kinetic Asymmetric Pauson–Khand Reaction of Allenyl Acetates
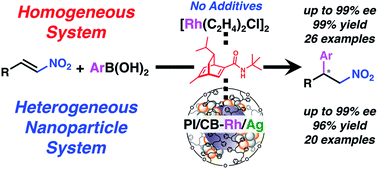
Rhodium-catalyzed asymmetric 1,4-addition reactions of aryl boronic acids with nitroalkenes: reaction mechanism and development of homogeneous and heterogeneous catalysts
DOI: 10.1039/C7SC03025H, Edge Article
 Open Access
Open Access   This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
  This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
Asymmetric 1,4-additions of arylboronic acids with nitroalkenes catalyzed by rhodium complexes or heterogeneous Rh-Ag bimetallic nanoparticles with a chiral diene ligand bearing a tertiary butyl amide moiety are developed.
The content of this RSS Feed (c) The Royal Society of Chemistry
Lewis Acid Enabled Copper-Catalyzed Asymmetric Synthesis of Chiral β-Substituted Amides
Rh-Catalyzed Conjugate Addition of Aryl and Alkenyl Boronic Acids to α-Methylene-β-lactones: Stereoselective Synthesis of trans-3,4-Disubstituted β-Lactones
N-heterocyclic Carbene–Cu-Catalyzed Enantioselective Conjugate Additions with Alkenylboronic Esters as Nucleophiles

Dynamic Kinetic Resolution in Rhodium-Catalyzed Asymmetric Arylation of Phospholene Oxides
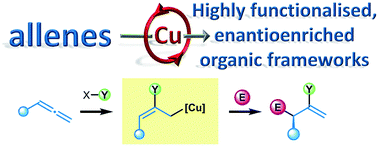
Enantioselective copper catalysed, direct functionalisation of allenes via allyl copper intermediates
DOI: 10.1039/C7SC01968H, Minireview
 Open Access
Open Access   This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
  This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
We explore the breadth of the copper catalysed functionalisation of allenes, which enables efficient access to enantioenriched, densely functionalised molecules.
The content of this RSS Feed (c) The Royal Society of Chemistry
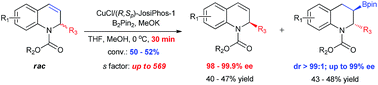
Kinetic resolution of racemic 2-substituted 1,2-dihydroquinolines via asymmetric Cu-catalyzed borylation
DOI: 10.1039/C7SC01556A, Edge Article
 Open Access
Open Access   This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
  This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
A highly efficient kinetic resolution of racemic 2-substituted 1,2-dihydroquinolines via asymmetric Cu-catalyzed borylation has been realized for the first time to afford chiral 3-boryl-1,2,3,4-tetrahydroquinolines and recover 2-substituted 1,2-dihydroquinolines with excellent enantioselectivities and selectivity factors of up to 569.
The content of this RSS Feed (c) The Royal Society of Chemistry
Regio- and stereospecific rhodium-catalyzed allylic alkylation with an acyl anion equivalent: an approach to acyclic [small alpha]-ternary [small beta],[gamma]-unsaturated aryl ketones
DOI: 10.1039/C6SC05705E, Edge Article
 Open Access
Open Access   This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
  This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
The regio- and stereospecific rhodium-catalyzed allylic alkylation of secondary allylic carbonates with cyanohydrin pronucleophiles facilitates the direct construction of acyclic [small alpha]-ternary [small beta],[gamma]-unsaturated aryl ketones.
The content of this RSS Feed (c) The Royal Society of Chemistry
Copper-Catalyzed, Stereoselective Cross-Coupling of Cyclic Allyl Boronic Acids with α-Diazoketones
Synthesis of Complex Tertiary Glycolates by Enantioconvergent Arylation of Stereochemically Labile α-Keto Esters
Rhodium-Catalyzed Hydrofunctionalization: Enantioselective Coupling of Indolines and 1,3-Dienes
Palladium-Catalyzed Asymmetric α-Arylation of Alkylnitriles
F– Nucleophilic-Addition-Induced Allylic Alkylation
Terminal-Selective Functionalization of Alkyl Chains by Regioconvergent Cross-Coupling
Abstract
Hydrocarbons are still the most important precursors of functionalized organic molecules, which has stirred interest in the discovery of new C−H bond functionalization methods. We describe herein a new step-economical approach that enables C−C bonds to be constructed at the terminal position of linear alkanes. First, we show that secondary alkyl bromides can undergo in situ conversion into alkyl zinc bromides and regioconvergent Negishi coupling with aryl or alkenyl triflates. The use of a suitable phosphine ligand favoring Pd migration enabled the selective formation of the linear cross-coupling product. Subsequently, mixtures of secondary alkyl bromides were prepared from linear alkanes by standard bromination, and regioconvergent cross-coupling then provided access to the corresponding linear arylation product in only two steps.

Remote control: Secondary alkyl bromides underwent regioconvergent Barbier–Negishi coupling with aryl triflates in the presence of a phosphine ligand favoring Pd migration to give linear products with good to high linear/branched selectivity (see scheme). This procedure was also applied to mixtures of secondary alkyl bromides, prepared from linear alkanes by standard bromination.
Enantioselective Palladium-Catalyzed Alkenylation of Trisubstituted Alkenols To Form Allylic Quaternary Centers
Enantioselective Formation of All-Carbon Quaternary Centers via C–H Functionalization of Methanol: Iridium-Catalyzed Diene Hydrohydroxymethylation
Enantioselective Pd-Catalyzed Allylic Alkylation Reactions of Dihydropyrido[1,2-a]indolone Substrates: Efficient Syntheses of (−)-Goniomitine, (+)-Aspidospermidine, and (−)-Quebrachamine
Abstract
The successful application of dihydropyrido[1,2-a]indolone (DHPI) substrates in Pd-catalyzed asymmetric allylic alkylation chemistry facilitates rapid access to multiple alkaloid frameworks in an enantioselective fashion. Strategic bromination at the indole C3 position greatly improved the allylic alkylation chemistry and enabled a highly efficient Negishi cross-coupling downstream. The first catalytic enantioselective total synthesis of (−)-goniomitine, along with divergent formal syntheses of (+)-aspidospermidine and (−)-quebrachamine, are reported herein.

Magnum (DH)PI: Application of dihydropyrido[1,2-a]indolone (DHPI) substrates in Pd-catalyzed asymmetric allylic alkylation chemistry enables rapid access to multiple alkaloid frameworks in an enantioselective fashion. The first catalytic enantioselective total synthesis of (−)-goniomitine, along with divergent formal syntheses of (+)-aspidospermidine and (−)-quebrachamine are reported.