Belén Rubial
Shared posts
Kinetic or Dynamic Control on a Bifurcating Potential Energy Surface? An Experimental and DFT Study of Gold-Catalyzed Ring Expansion and Spirocyclization of 2-Propargyl-β-tetrahydrocarbolines
Homogeneous Gold-Catalyzed Cyclization Reactions of Alkynes with N- and S-Nucleophiles
Abstract
This review covers the formation of N- and S-containing heterocycles, initiated by gold-catalyzed nucleophilic attack of N- or S-nucleophiles onto alkynes. These types of nucleophiles have been somewhat overlooked as compared to their C- or O-counterparts in other reviews. In this particular work, their intramolecular gold-mediated attack onto alkynes is reviewed in depth. It is structured in such a fashion that the reader will get a clear view of which substrates react in which cyclization mode.

Oxidant-Free Au(I)-Catalyzed Halide Exchange and Csp2–O Bond Forming Reactions
One-Step Conversion of Methyl Ketones to Acyl Chlorides
Route to α-Aryl Phosphonoacetates: Useful Synthetic Precursors in the Horner–Wadsworth–Emmons Olefination
Dinuclear planar chiral ferrocenyl gold(I) & gold(II) complexes
DOI: 10.1039/C5CC07018J, Communication
The first scalemic ferrocenyl gold(I) and gold(II) complexes have been prepared and structurally analysed by X-ray, (spectro)electrochemical and DFT studies.
The content of this RSS Feed (c) The Royal Society of Chemistry
Stereodivergent Dual Catalytic α-Allylation of Protected α-Amino- and α-Hydroxyacetaldehydes
Abstract
Fully stereodivergent dual-catalytic α-allylation of protected α-amino- and α-hydroxyacetaldehydes is achieved through iridium- and amine-catalyzed substitution of racemic allylic alcohols with chiral enamines generated in situ. The operationally simple method furnishes useful aldehyde building blocks in good yields, more than 99 % ee, and with d.r. values greater than 20:1 in some cases. Additionally, the γ,δ-unsaturated products can be further functionalized in a stereodivergent fashion with high selectivity and with preservation of stereochemical integrity at the Cα position.

Be selective: Ir-catalyzed allylic substitution of racemic allylic alcohols with chiral enamines generated in situ enables the fully stereodivergent α-allylation of protected α-amino- and α-hydroxyacetaldehydes. The method furnishes α-N/O-substituted γ,δ-unsaturated aldehydes in good yields and with excellent enantioselectivities. Phth=phthalimide; cod=1,5-cyclooctadiene; Ts=p-toluenesulfonyl; DCE=1,2-dichloroethane.
Synthesis and Evaluation of 5,5′-Bitetralone-Based Chiral Phosphoric Acids
Synthesis of 2,5-Dihydrofurans via a Gold(I)-Catalyzed Formal [4 + 1] Cycloaddition of α-Diazoesters and Propargyl Alcohols
[Report] Conversion of alcohols to enantiopure amines through dual-enzyme hydrogen-borrowing cascades
[Report] O–H hydrogen bonding promotes H-atom transfer from α C–H bonds for C-alkylation of alcohols
Synthesis and Catalytic Use of Gold(I) Complexes Containing a Hemilabile Phosphanylferrocene Nitrile Donor
Abstract
Removal of the chloride ligand from [AuCl(1-κP)] (2) containing a P-monodentate 1′-(diphenylphosphanyl)-1-cyanoferrocene ligand (1), by using silver(I) salts affords cationic complexes of the type [Au(1)]X, which exist either as cyclic dimers [Au(1)]2X2 (3 a, X=SbF6; 3 c, X=NTf2) or linear coordination polymers [Au(1)]nXn (3 a′, X=SbF6; 3 b′, X=ClO4), depending on anion X and the isolation procedure. As demonstrated for 3 a′, the polymers can be readily cleaved by the addition of donors, such as Cl−, tetrahydrothiophene (tht) or 1, giving rise to the parent compound 2, [Au(tht)(1-κP)][SbF6] (5 a) or [Au(1-κP)2][SbF6] (4 a), respectively, of which the last two compounds can also be prepared by stepwise replacement of tht in [Au(1-κP)2][SbF6]. The particular combination of a firmly coordinated (phosphane) and a dissociable (nitrile) donor moieties renders complexes 3/3′ attractive for catalysis because they can serve as shelf-stable precursors of coordinatively unsaturated AuI fragments, analogous to those that result from the widely used [Au(PR3)(RCN)]X catalysts. The catalytic properties of the Au-1 complexes were evaluated in model annulation reactions, such as the synthesis of 2,3-dimethylfuran from (Z)-3-methylpent-2-en-4-yn-1-ol and oxidative cyclisation of alkynes with nitriles to produce 2,5-disubstituted 1,3-oxazoles. Of the compounds tested (2, 3 a′, 3 b′, 3 a, 4 a and 5 a), the best results were consistently achieved with dimer 3 c, which has good solubility in organic solvents and only one firmly bound donor at the gold atom. This compound was advantageously used in the key steps of annuloline and rosefuran syntheses.

Self-stabilised catalysts: Dimeric and polymeric gold(I) complex cations with 1′-(diphenylphosphanyl)-1-cyanoferrocene ligand (L) coordinated as a hemilabile P,N donor, [Au(L-κ2P,N)]+, readily undergo cleavage of the Au![[BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) CN coordination bond and can be thus advantageously used as precursors for highly catalytically active, silver-free AuI species (see figure).
CN coordination bond and can be thus advantageously used as precursors for highly catalytically active, silver-free AuI species (see figure).
Insights into the Gold-Catalyzed Propargyl Ester Rearrangement/Tandem Cyclization Sequence: Radical versus Gold Catalysis—Myers–Saito- versus Schmittel-Type Cyclization
Abstract
A detailed study of the gold-catalyzed tandem 1,3-carboxy migration/allene–enyne cycloisomerization was undertaken. It was found that after the initial allene formation the selectivity of the reaction is strongly influenced by the polarization of the remaining alkyne. Depending on the substitution pattern of the starting diynes, either a Schmittel- or a Myers–Saito-type cyclization was triggered. The 6-endo-dig Myers–Saito-type cyclization gave access to benzo[b]fluorenes, while the Schmittel pathway (5-exo-dig) delivered benzofulvenes as final products. In special cases a yet unknown pathway was opened by the ambiphilic nature of the allene moiety. In these cases completely different products were obtained by the nucleophilic attack of the alkyne moiety onto the allene that can also act as an electrophile. Mechanistic studies revealed that diradical pathways can be ruled out for this type of tandem cyclization reactions and it is shown that both steps of the reaction cascade are catalyzed by the gold complex.

Substitution patterns: A detailed study of the gold-catalyzed tandem 1,3-carboxy migration/allene–enyne cycloisomerization was undertaken. It was found that the selectivity of the reaction is strongly influenced by the polarization of the remaining alkyne. Depending on the substitution pattern of the starting diynes, either a Schmittel- or a Myers–Saito-type cyclization was triggered (see scheme).
Stereo- and Chemodivergent NHC-Promoted Functionalisation of Arylalkylketenes with Chloral
Abstract
Stereo- and chemodivergent enantioselective reaction pathways are observed upon treatment of alkylarylketenes and trichloroacetaldehyde (chloral) with N-heterocyclic carbenes, giving selectively either β-lactones (up to 88:12 dr, up to 94 % ee) or α-chloroesters (up to 94 % ee). Either 2-arylsubstitution or an α-branched iPr alkyl substituent within the ketene favours the chlorination pathway, allowing chloral to be used as an electrophilic chlorinating reagent in asymmetric catalysis.

Taming chloral two ways: Stereo- and chemodivergent enantioselective reaction pathways are observed upon treatment of alkylarylketenes and trichloroacetaldehyde (chloral) with N-heterocyclic carbenes (NHCs), giving selectively either β-lactones (up to 88:12 dr, up to 94 % ee) or α-chloroesters (up to 94 % ee). Either 2-arylsubstitution or an α-branched iPr alkyl substituent within the ketene favours the chlorination pathway (see scheme).
Synthesis of Chiral Pyrazoles: A 1,3-Dipolar Cycloaddition/[1,5] Sigmatropic Rearrangement with Stereoretentive Migration of a Stereogenic Group
Abstract
The reactions between terminal alkynes and α-chiral tosylhydrazones lead to the obtention of chiral pyrazoles with a stereogenic group directly attached at a nitrogen atom. The cascade reaction includes decomposition of the hydrazone into a diazocompound, 1,3-dipolar cycloaddition of the diazo compound with the alkyne, and [1,5] sigmatropic rearrangement with migration of the stereogenic group. This strategy has been successfully applied to the synthesis of structurally diverse chiral pyrazoles through α-chiral tosylhydrazones, obtained from α-phenylpropionic acid, α-amino acids, and 2-methoxycyclohexanone. Notably, the stereoretention of the [1,5] sigmatropic rearrangements represent very rare examples of this stereospecific transformation.

Maintaining a (con)figur(ation): Retention of configuration in [1,5] sigmatropic shifts are predicted by the Woodward–Hoffmann rules, but are not well-documented experimentally. Such shifts are observed in the reactions of α-chiral tosylhydrazones with terminal alkynes. The chiral pyrazole products are formed by a 1,3-dipolar cycloaddition and subsequent site-, regio-, and stereospecific [1s,5s] sigmatropic rearrangement. Ts=4-toluenesulfonyl.
Highly Regio-, Diastereo-, and Enantioselective Gold(I)-Catalyzed Intermolecular Annulations with N-Allenamides at the Proximal CC Bond
Abstract
A highly enantioselective gold(I)-catalyzed intermolecular annulation of 2-(1-alkynyl)-2-alken-1-ones with N-allenamides is presented. The present work represents the first example of a gold-catalyzed annulation with the proximal C![[DOUBLE BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8fe.gif) C bond of an N-allenamide, and is distinctly different from the previously observed annulations at the distal C
C bond of an N-allenamide, and is distinctly different from the previously observed annulations at the distal C![[DOUBLE BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8fe.gif) C bond. Interestingly, both enantiomers of the products could be obtained in good yields with high regio-, diastereo-, and enantioselectivity by using either diastereomer of a binol-derived phosphoramidite as a chiral ligand.
C bond. Interestingly, both enantiomers of the products could be obtained in good yields with high regio-, diastereo-, and enantioselectivity by using either diastereomer of a binol-derived phosphoramidite as a chiral ligand.

On the double: An unprecedented [3+2] annulation takes place with the proximal C![[DOUBLE BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8fe.gif) C bond of N-allenamides in the presence of a chiral gold catalyst. Both enantiomers could be obtained in good yields with high regio-, diastereo-, and enantioselectivities by using either diastereomer of a binol-derived phosphoramidite ligand. binol=1,1′-bi-2-naphthol.
C bond of N-allenamides in the presence of a chiral gold catalyst. Both enantiomers could be obtained in good yields with high regio-, diastereo-, and enantioselectivities by using either diastereomer of a binol-derived phosphoramidite ligand. binol=1,1′-bi-2-naphthol.
Gold(I)-Catalyzed Addition of Silylacetylenes to Acylsilanes: Synthesis of Indanones by CH Functionalization through a Gold(I) Carbenoid
Abstract
A gold(I)-catalyzed synthesis of indanones from trimethylsilylacetylenes and acylsilanes is presented. The reaction is initiated through a synergistic acylsilane activation–gold acetylide formation and involves consecutive alkyne σ-gold(I) addition, π-activation, and 1,2-migration of a silyl group. Studies performed on the reaction mechanism allowed to establish the nature of the silyl migrating group and invoke the participation of a gold(I) carbenoid intermediate. The reaction is completed by a gold(I) C![[BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) H functionalization step.
H functionalization step.

The elegant way: A gold(I)-catalyzed synthesis of indanones from trimethylsilylacetylenes and acylsilanes was developed. The reaction involves a synergistic acylsilane activation–gold acetylide formation and consecutive alkyne σ-gold(I) addition, π-activation, and 1,2-silyl migration. Mechanistic studies suggest the participation of a gold(I) carbenoid intermediate.
Mechanistic Understanding of the Divergent Cyclizations of o-Alkynylbenzaldehyde Acetals and Thioacetals Catalyzed by Metal Halides
Abstract
The mechanisms of regiodivergent cyclizations of o-alkynylbenzaldehyde acetals and thioacetals catalyzed by Pd and Pt halides are studied. DFT calculations found that both reactions are initiated by electrophilic activation of the acetylenic moiety instead of the previously proposed metal-triggered C![[BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) X (X=O, S) cleavage. Both the regioselective cyclization of the π-alkyne complex and the chemoselective [1,2]-migration in the carbenoid intermediate were determined as key steps to achieving the observed divergence. For acetal derivatives containing an internal alkyne, the 6-endo-dig cyclization is more favorable and leads to the carbenoid intermediate easily through further steps of C
X (X=O, S) cleavage. Both the regioselective cyclization of the π-alkyne complex and the chemoselective [1,2]-migration in the carbenoid intermediate were determined as key steps to achieving the observed divergence. For acetal derivatives containing an internal alkyne, the 6-endo-dig cyclization is more favorable and leads to the carbenoid intermediate easily through further steps of C![[BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) X fragmentation and carbocation cyclization. Then, from the carbenoid intermediate, the [1,2]-migration of sulfur is easier than that of H, Me, and Ph; whereas, a reversed aptitude was predicted for the oxygen analogue, which is consistent with the greater ability of sulfur atoms to stabilize β-carbocations. However, for precursors containing a terminal alkyne, the 5-exo-dig pathway is preferred and only the 1,2-disubstituted indene product is seen, irrespective of the nature of the acetal; thus, a different product from that reported in the literature is predicted for benzaldehyde acetal with a terminal alkyne at the ortho position. This prediction led us to reconsider some of the reported results and hidden realities were uncovered with solid new experimental evidence.
X fragmentation and carbocation cyclization. Then, from the carbenoid intermediate, the [1,2]-migration of sulfur is easier than that of H, Me, and Ph; whereas, a reversed aptitude was predicted for the oxygen analogue, which is consistent with the greater ability of sulfur atoms to stabilize β-carbocations. However, for precursors containing a terminal alkyne, the 5-exo-dig pathway is preferred and only the 1,2-disubstituted indene product is seen, irrespective of the nature of the acetal; thus, a different product from that reported in the literature is predicted for benzaldehyde acetal with a terminal alkyne at the ortho position. This prediction led us to reconsider some of the reported results and hidden realities were uncovered with solid new experimental evidence.

Where paths diverge: In-depth mechanistic understanding of the regiodivergent cyclizations of o-alkynylbenzaldehyde acetals and thioacetals catalyzed by Pd and Pt halides is achieved by combined theoretical and experimental studies (see scheme).
Formal Gold-to-Gold Transmetalation of an Alkynyl Group Mediated by Palladium: A Bisalkynyl Gold Complex as a Ligand to Palladium
Abstract
The reaction of [Au(C![[TRIPLE BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8fd.gif) C
C![[BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) n-Bu)]n with [Pd(η3-allyl)Cl(PPh3)] results in a ligand and alkynyl rearrangement, and leads to the heterometallic complex [Pd(η3-allyl){Au(C
n-Bu)]n with [Pd(η3-allyl)Cl(PPh3)] results in a ligand and alkynyl rearrangement, and leads to the heterometallic complex [Pd(η3-allyl){Au(C![[TRIPLE BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8fd.gif) C
C![[BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) n-Bu)2}]2 (3) with an unprecedented bridging bisalkynyl–gold ligand coordinated to palladium. This is a formal gold-to-gold transmetalation that occurs through reversible alkynyl transmetalations between gold and palladium.
n-Bu)2}]2 (3) with an unprecedented bridging bisalkynyl–gold ligand coordinated to palladium. This is a formal gold-to-gold transmetalation that occurs through reversible alkynyl transmetalations between gold and palladium.

Back and forth! Direct gold-to-palladium and reverse palladium-to-gold alkynyl transmetalations effectively produce a deceptively simple gold-to-gold transfer. The resulting bisalkynyl–gold complexes act as bridging ligands to palladium atoms in an unprecedented structural motive (see scheme).
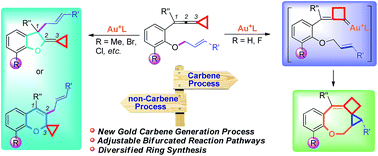
Gold(I)-catalyzed cycloisomerization of vinylidenecyclopropane-enes via carbene or non-carbene processes
DOI: 10.1039/C5SC01806D, Edge Article
 Open Access
Open Access   This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
  This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
Gold catalyzed cycloisomerization of aromatic ring tethered vinylidenecyclopropane-enes provides a divergent synthetic protocol for the construction of O-containing fused heterocycles through controllable carbene or non-carbene related processes.
The content of this RSS Feed (c) The Royal Society of Chemistry
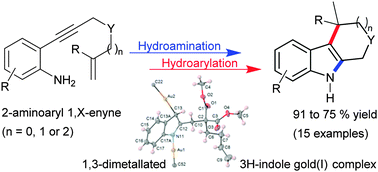
Gold(I)-catalysed cascade reactions in the synthesis of 2,3-fused indole derivatives
DOI: 10.1039/C5CC04606H, Communication
Unactivated alkenes and 1,3-unsubstituted indoles in gold(I)-catalysed hydroaminative/arylative cascade cyclizations.
The content of this RSS Feed (c) The Royal Society of Chemistry
Highly Regio- and Diastereoselective Formation of Tetrasubstituted (Z)-1,2-Dihaloalkenes from the Halogenation of Trimethylsilyl Alkynes with ICl
A Theoretical Study on the Protodeauration Step of the Gold(I)-Catalyzed Organic Reactions
An Au(I)-catalyzed rearrangement/cyclization cascade toward the synthesis of 2-substituted-1,4,5,6-tetrahydrocyclopenta[b]pyrrole
DOI: 10.1039/C5CC03979G, Communication
An Au(I)-catalyzed tandem reaction, involving a Meyer-Schuster rearrangement/1,2-migration/Paal-Knorr cyclization cascade has been successfully developed.
The content of this RSS Feed (c) The Royal Society of Chemistry
Catalyst-Dependent Stereodivergent and Regioselective Synthesis of Indole-Fused Heterocycles through Formal Cycloadditions of Indolyl-Allenes
Gold(I)-Catalyzed 1,3-O-Transposition Reactions: Ynesulfonamides to Ynamides
Abstract
The gold-catalyzed 1,3-O-transposition reaction of ynesulfonamides provides a practical synthetic protocol for the synthesis of ynamides under mild conditions. This is the first 1,3-O-transposition example of ynesulfonamides in which a heteroatom is attached to the alkynyl terminal. A plausible mechanism is been proposed on the basis of 18O-labeling and control experiments as well as DFT calculation. By simple treatment, the obtained ynamides can easily be transformed into useful products.

The gold-catalyzed 1,3-O-transposition reaction of ynesulfonamides provides a practical synthetic protocol for the synthesis of ynamides under mild conditions. This is the first 1,3-O-transposition example of ynesulfonamides in which a heteroatom is attached to the alkynyl terminal.
Silica-Supported Cationic Gold(I) Complexes as Heterogeneous Catalysts for Regio- and Enantioselective Lactonization Reactions
Chiral Brønsted Acid as a True Catalyst: Asymmetric Mukaiyama Aldol and Hosomi–Sakurai Allylation Reactions
Synthesis and Characterization of a Gold Vinylidene Complex Lacking π-Conjugated Heteroatoms
Abstract
Hydride abstraction from the gold (disilyl)ethylacetylide complex [(P)Au{η1-C![[TRIPLE BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8fd.gif) CSi(Me)2CH2CH2SiMe2H}] (P=P(tBu)2o-biphenyl) with triphenylcarbenium tetrakis(pentafluorophenyl)borate at −20 °C formed the cationic gold (β,β-disilyl)vinylidene complex [(P)Au
CSi(Me)2CH2CH2SiMe2H}] (P=P(tBu)2o-biphenyl) with triphenylcarbenium tetrakis(pentafluorophenyl)borate at −20 °C formed the cationic gold (β,β-disilyl)vinylidene complex [(P)Au![[DOUBLE BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8fe.gif) C
C![[DOUBLE BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8fe.gif) CSi(Me)2CH2CH2Si(Me)2]+B(C6F5)4− with ≥90 % selectivity. 29Si NMR analysis of this complex pointed to delocalization of positive charge onto both the β-silyl groups and the (P)Au fragment. The C1 and C2 carbon atoms of the vinylidene complex underwent facile interconversion (ΔG≠=9.7 kcal mol−1), presumably via the gold π-disilacyclohexyne intermediate [(P)Au{η2-C
CSi(Me)2CH2CH2Si(Me)2]+B(C6F5)4− with ≥90 % selectivity. 29Si NMR analysis of this complex pointed to delocalization of positive charge onto both the β-silyl groups and the (P)Au fragment. The C1 and C2 carbon atoms of the vinylidene complex underwent facile interconversion (ΔG≠=9.7 kcal mol−1), presumably via the gold π-disilacyclohexyne intermediate [(P)Au{η2-C![[TRIPLE BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8fd.gif) CSi(Me)2CH2CH2Si(Me)2}]+B(C6F5)4−.
CSi(Me)2CH2CH2Si(Me)2}]+B(C6F5)4−.

Good as gold: Cationic gold (β,β-disilyl)vinylidene complex 1 was generated by addition of a pendant silylium ion to the C![[TRIPLE BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8fd.gif) C bond of a gold acetylide complex (see scheme, P=PtBu2(o-biphenyl)). The vinylidene C1 and C2 atoms of 1 undergo facile interconversion, presumably via a π-disilacyclohexyne intermediate. 29Si NMR analysis of 1 indicates delocalization of positive charge onto both the β-silyl groups and the (P)Au fragment.
C bond of a gold acetylide complex (see scheme, P=PtBu2(o-biphenyl)). The vinylidene C1 and C2 atoms of 1 undergo facile interconversion, presumably via a π-disilacyclohexyne intermediate. 29Si NMR analysis of 1 indicates delocalization of positive charge onto both the β-silyl groups and the (P)Au fragment.
The Stabilizing Effects in Gold Carbene Complexes
Abstract
Bonding and stabilizing effects in gold carbene complexes are investigated by using Kohn–Sham density functional theory (DFT) and the intrinsic bond orbital (IBO) approach. The π-stabilizing effects of organic substituents at the carbene carbon atom coordinated to the gold atom are evaluated for a series of recently isolated and characterized complexes, as well as intermediates of prototypical 1,6-enyne cyclization reactions. The results indicate that these effects are of particular importance for gold complexes especially because of the low π-backbonding contribution from the gold atom.

Is anybody there? Intrinsic bond orbital analyses based on DFT calculations on gold carbene complexes indicate little π backbonding from gold (A) and π stabilization from organic fragments even in cyclopropyl-substituted gold carbene complex intermediates (B, C).