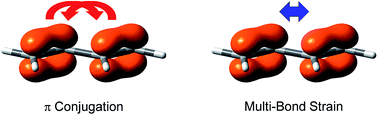Michael Cowley
Shared posts
[ASAP] Copper-Catalyzed C–B(sp3) Bond Formation through the Intermediacy of Cu–B(sp3) Complex
[ASAP] IPr*diNHC: Sterically Adaptable Dinuclear N-Heterocyclic Carbenes

Exploring frustrated radical pairs through the persistent radical effect: methods of generation and recent applications
DOI: 10.1039/D5CC00946D, Highlight
 Open Access
Open Access   This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
  This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
This highlight centers on the role of the persistent radical effect in frustrated radical pairs (FRPs). Three ways of generating FRPs via FLPs, including single electron transfer and substrate-assisted methods are described.
The content of this RSS Feed (c) The Royal Society of Chemistry
These are the most-cited research papers of all time
Nature, Published online: 15 April 2025; doi:10.1038/d41586-025-01124-w
Some studies have received hundreds of thousands of citations, Nature’s updated analysis shows.Science’s golden oldies: the decades-old research papers still heavily cited today
Nature, Published online: 15 April 2025; doi:10.1038/d41586-025-01126-8
An analysis for Nature reveals the studies that appear most in the reference lists of current publications.Exclusive: the most-cited papers of the twenty-first century
Nature, Published online: 15 April 2025; doi:10.1038/d41586-025-01125-9
A Nature analysis reveals the 25 highest-cited papers published this century and explores why they are breaking records.Ensuring that conferences are inclusive requires diverse organizers
Nature, Published online: 15 April 2025; doi:10.1038/d41586-025-01204-x
Ensuring that conferences are inclusive requires diverse organizers[ASAP] Origin of Intramolecular versus Intermolecular C–H Arene Activation Selectivity by Cyclopentadienyl–Triphenylphosphine Iridium

Reversible addition of ethene to gallium(I) monomers and dimers
DOI: 10.1039/D4SC06894G, Edge Article
 Open Access
Open Access   This article is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported Licence.
  This article is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported Licence.
Transient or stable digallenes react reversibly with ethene. Such reversibility is a hallmark of transition metal chemistry but remains uncommon for low-valent p-block compounds.
The content of this RSS Feed (c) The Royal Society of Chemistry
Direct Conversion of Aromatic Lactones into Bioisosteres by Carbonyl‐to‐Boranol Exchange

A nickel-catalyzed group-exchange strategy has been developed for the direct conversion of aromatic lactones into cyclic hemiboronic acid bioisosteres. Scope evaluation and product derivatization experiments demonstrate broad functional-group compatibility and the synthetic value of this strategy. Furthermore, the application of this methodology to the rapid modification of lactone cores in bioactive molecules underscores its practical utility.
Abstract
Bioisosteric replacement is an important strategy in drug discovery and is commonly practiced in medicinal chemistry; however, the incorporation of bioisosteres typically requires laborious multistep de novo synthesis. The direct conversion of a functional group into its corresponding bioisostere is of particular significance in evaluating structure-property relationships. Herein, we report a functional-group-exchange strategy that enables the direct conversion of aromatic lactones, a prevalent motif in bioactive molecules, into their corresponding cyclic hemiboronic acid bioisosteres. Scope evaluation and product derivatization experiments demonstrate the synthetic value and broad functional-group compatibility of this strategy, while the application of this methodology to the rapid remodeling of chromenone cores in bioactive molecules highlights its utility.
The evolution of machine learning potentials for molecules, reactions and materials
DOI: 10.1039/D5CS00104H, Review Article
This review offers a comprehensive overview of the development of machine learning potentials for molecules, reactions, and materials over the past two decades, evolving from traditional models to the state-of-the-art.
The content of this RSS Feed (c) The Royal Society of Chemistry
Synthesis and reactivity of a parent phosphathioethynolato-borane and a boraarsaketene
DOI: 10.1039/D5SC01996F, Edge Article
 Open Access
Open Access   This article is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported Licence.
  This article is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported Licence.
The first main group phosphathioethynolates, [LBH2(SCP)] (L = carbene), combine phosphaalkyne, sulfide and hydroborane reactivity to generate complex B,P,S-polyheterocycles, while [LBH2(AsCO)] leads to a rare arsinidene–alkene π complex.
The content of this RSS Feed (c) The Royal Society of Chemistry
Probing the reactivity of a transient Al(I) species with substituted arenes
DOI: 10.1039/D4CC03904A, Communication
 Open Access
Open Access   This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
  This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
A series of rare dialumene-arene adducts have been isolated and their ‘masked’ reactivity as a source of Al(I) probed.
The content of this RSS Feed (c) The Royal Society of Chemistry
[ASAP] Mixed-Valence Iron Complexes Containing End-On Bridging Cyaphide Ions

Nucleophilic Triphenylgermanides of Magnesium and Calcium

Reactions of the dimeric alkaline earth hydrides, [(BDI)AeH]2 (BDI=HC{(Me)CNDipp}2; Dipp=2,6-i-Pr2C6H3; Ae=Mg, Ca), with Ph3GeH in the presence of THF provide H2 and [(BDI)AeGePh3(THF)]. Although the synthesis of the magnesium derivative is complicated by THF ring opening, the calcium reagent is a viable source of the triphenylgermanide nucleophile.
Abstract
The dimeric calcium and magnesium hydrides, [(BDI)AeH]2 [BDI=HC{(Me)CNDipp}2, Dipp=2,6-i-Pr2C6H3; Ae=Mg or Ca] do not react with Ph3GeH in non-coordinating solvent. Addition of THF, however, induces deprotonation and access to monomeric Ae-germanide complexes, [(BDI)Ae{GePh3}(THF)], both of which have been structurally characterized. Although this process is facile when Ae=Ca, the analogous magnesium-based reaction requires heating to temperatures >100 °C, under which conditions germanide formation is complicated by THF ring opening and the generation of an alkaline earth germyl-C-terminated n-butoxide, [(BDI)Mg{μ2-O(CH2)4GePh3}]. Reactions of [(BDI)Ca{GePh3}(THF)] with N,N′-di-isopropylcarbodiimide and benzophenone provide the respective germylamidinate and germylalkoxide derivatives, [(BDI)Ca{(i-PrN)2CGePh3}(THF)] and [(BDI)Ca{OC(GePh3)Ph2}(THF)], demonstrating its potential as a well-defined and soluble source of the [Ph3Ge]− anion in nucleophilic addition reactions.
Hydridostannylene Derivatives of Magnesium and Calcium

Reactions of [(2,6-Dipp2C6H3)Sn(μ-H)]2 (Dipp=2,6-i-Pr2C6H3) with [(BDI)AeH]2 (BDI=HC{(Me)CNDipp}2; Ae=Mg, Ca) provide heterobimetallic bis-μ2-hydrido species, [[(BDI)Ae-μ-(H)2-Sn(C6H3-2,6-Dipp2)]. Although both compounds react with hex-1-ene to provide alkaline earth derivatives of tetravalent organostannides, the magnesium reagent promotes twofold addition of the alkene whereas the calcium-based reaction is arrested after a single hydrostannylation event.
Abstract
Reactions of a m-terphenylhydridostannylene with β-diketiminato magnesium and calcium hydrides provide bis-μ2-hydrido species, the heterobimetallic constitutions of which are maintained after the addition of THF donor solvent. In both cases, reactions with hex-1-ene result in the formation of tetravalent organostannyl alkaline earth derivatives. Whereas the magnesium reagent undergoes facile twofold addition, the calcium-centered process is arrested after a single alkene reduction event. This contrasting behavior is assessed to result from the heavier alkaline earth element's ability to form a persistent polyhapto interaction with an arene substituent of the m-terphenyl ligand.
[ASAP] Facile, Reversible Hydrogen Activation by Low-Coordinate Magnesium Oxide Complexes

[ASAP] Methane Beryllation Catalyzed by a Base Metal Complex

[ASAP] Cooperative Heterobimetallic CO2 Activation Involving a Mononuclear Aluminum(II) Intermediate

[ASAP] Unlocking a Diazirine Long-Lived Nuclear Singlet State via Photochemistry: NMR Detection and Lifetime of an Unstabilized Diazo-Compound
Direct access to a cAAC-supported dihydrodiborene and its dianion
Michael Cowleynice
DOI: 10.1039/C8CC01580E, Communication
The stepwise reduction of cyclic (alkyl)(amino)carbene-supported (dihalo)hydroboranes provides access to a highly sensitive (halo)hydroboryl anion, followed by a dihydrodiborene and, finally, a bis(alkylidene-borane) dianion.
The content of this RSS Feed (c) The Royal Society of Chemistry
Synthesis and Catalytic Reactivity of Bis(amino)cyclopropenylidene Carbene–Borane Adducts
A Non-Exploding Alkali Metal Drop on Water: From Blue Solvated Electrons to Bursting Molten Hydroxide
Abstract
Alkali metals in water are always at the brink of explosion. Herein, we show that this vigorous reaction can be kept in a non-exploding regime, revealing a fascinating richness of hitherto unexplored chemical processes. A combination of high-speed camera imaging and visible/near-infrared/infrared spectroscopy allowed us to catch and characterize the system at each stage of the reaction. After gently placing a drop of a sodium/potassium alloy on water under an inert atmosphere, the production of solvated electrons became so strong that their characteristic blue color could be observed with the naked eye. The exoergic reaction leading to the formation of hydrogen and hydroxide eventually heated the alkali metal drop such that it became glowing red, and part of the metal evaporated. As a result of the reaction, a perfectly transparent drop consisting of molten hydroxide was temporarily stabilized on water through the Leidenfrost effect, bursting spectacularly after it had cooled sufficiently.

The vigorous reaction of alkali metals in water was kept in a non-exploding regime through gently placing a drop of Na/K alloy on water. Initially, solvated electrons are produced (blue color), before the exoergic reaction leading to H2 and OH− heats the drop such that it becomes red, and part of the metal evaporates. A transparent drop of molten hydroxide is then temporarily stabilized on the water through the Leidenfrost effect before bursting spectacularly.
Synthesis and Structure of Bis(diphenylphosphinimino)methanide and Bis(diphenylphosphinimino)methanediide Beryllium Complexes
Marriage of heavy main group elements with [small pi]-conjugated materials for optoelectronic applications
DOI: 10.1039/C6CC04023C, Feature Article
This review showcases recent work devoted to placing heavy inorganic elements within [small pi]-conjugated frameworks and the novel properties that can arise.
The content of this RSS Feed (c) The Royal Society of Chemistry
Intramolecular multi-bond strain: the unrecognized side of the dichotomy of conjugated systems
DOI: 10.1039/C6SC00454G, Edge Article
 Open Access
Open Access   This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
  This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
Apart from the more familiar [small pi]-conjugation, there is also significant [small pi]-[small pi] repulsion which is a kind of unrecognized intramolecular strain and can be quantified with the linear B4H2 model system.
The content of this RSS Feed (c) The Royal Society of Chemistry
4-Position-Selective C–H Perfluoroalkylation and Perfluoroarylation of Six-Membered Heteroaromatic Compounds
Alkali Metal Carbenoids: A Case of Higher Stability of the Heavier Congeners
Abstract
As a result of the increased polarity of the metal–carbon bond when going down the group of the periodic table, the heavier alkali metal organyl compounds are generally more reactive and less stable than their lithium congeners. We now report a reverse trend for alkali metal carbenoids. Simple substitution of lithium by the heavier metals (Na, K) results in a significant stabilization of these usually highly reactive compounds. This allows their isolation and handling at room temperature and the first structure elucidation of sodium and potassium carbenoids. The control of stability was used to control reactivity and selectivity. Hence, the Na and K carbenoids act as selective carbene-transfer reagents, whereas the more labile lithium systems give rise to product mixtures. Additional fine tuning of the M−C interaction by means of crown ether addition further allows for control of the stability and reactivity.

Heavy but stable: The reactivity of s-block metal organyl compounds generally increases when going down the group of the periodic table, often limiting applications of the heavier congeners. However, the reverse of this trend has now been demonstrated for alkali metal carbenoids by replacement of Li by Na or K. The resulting compounds exhibit an increased thermal stability, which allows their isolation and selective application.
Can main group systems act as superior catalysts for dihydrogen generation reactions? A computational investigation
Michael Cowleygood pone to share Steph. Look at the compound they propose in this paper... they are bonkers! First one to propose a synthetic route to one of them wins... something (the chance to make it?)
DOI: 10.1039/C5DT01058F, Paper
The density functional theory (DFT) calculations reveal the potential of newly proposed main group germanium hydride systems to effect important chemical transformations, such as the catalytic cleavage of the O-H bond in water and alcohols, with significantly greater efficiency than the existing, state-of-the-art post-transition metal based systems.
The content of this RSS Feed (c) The Royal Society of Chemistry