Cuijingjing680
Shared posts
Remarkable Magnetic Coupling Interactions in Multi-Beryllium-Expanded Small Graphene-like Molecules with Well-Defined Polyradical Characters
Enantioselective preparation and chemoselective cross-coupling of 1,1-diboron compounds
Nature Chemistry 5, 634 (2013). doi:10.1038/nchem.1679
Author: Jack Chang Hung Lee, Robert McDonald & Dennis G. Hall
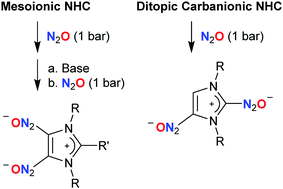
Fixation of nitrous oxide by mesoionic and carbanionic N-heterocyclic carbenes
DOI: 10.1039/C7CC01592E, Communication
Double fixation of laughing gas (N2O) can be achieved under mild conditions using mesoionic or ditopic carbanionic carbenes.
The content of this RSS Feed (c) The Royal Society of Chemistry
Synthesis of a distinct water dimer inside fullerene C70

Nature Chemistry. doi:10.1038/nchem.2464
Authors: Rui Zhang, Michihisa Murata, Tomoko Aharen, Atsushi Wakamiya, Takafumi Shimoaka, Takeshi Hasegawa & Yasujiro Murata
Endohedral C70 fullerenes containing either one or two water molecules have now been prepared using a molecular-surgery approach. The structure of H2O@C70 was determined by single-crystal X-ray analysis, revealing the encapsulated water molecule to be in an off-centre position. In (H2O)2@C70, the two water molecules form a discrete dimer held together with a single hydrogen bond.
Boron-Catalyzed Aromatic C–H Bond Silylation with Hydrosilanes
Cuijingjing680why not reported by someone who's major lies in main group chemistry
[Report] A bacterium that degrades and assimilates poly(ethylene terephthalate)
Cuijingjing680I am not doing Bio or Environment science, but this paper is really fantastic to me.
Polyboramines for Hydrogen Release: Polymers Containing Lewis Pairs in their Backbone
Cuijingjing680polymer, hydrogen storage
Abstract
The one-step polycondensation of diamines and diboranes triggered by the in situ deprotonation of the diammonium salts and concomitant reduction of bisboronic acids leads to the assembly of polymer chains through multiple Lewis pairing in their backbone. These new polyboramines are dihydrogen reservoirs that can be used for the hydrogenation of imines and carbonyl compounds. They also display a unique dihydrogen thermal release profile that is a direct consequence of the insertion of the amine–borane linkages in the polymeric backbone.

Polybor-a-mine of H2: The one-step polycondensation of diamine and diborane precursors leads to polymer chains assembled by multiple Lewis pairs (see scheme). The new polyboramines are dihydrogen reservoirs that can hydrogenate imines, aldehydes, and ketones. The polyboramines release H2 thermally with a unique profile, which is a direct consequence of the polymeric nature of the material.
Sex redefined
Cuijingjing680So you are man (woman), are you sure?
Sex redefined
Nature 518, 7539 (2015). http://www.nature.com/doifinder/10.1038/518288a
Author: Claire Ainsworth
The idea of two sexes is simplistic. Biologists now think there is a wider spectrum than that.