Shared posts
[ASAP] Dual Roles for Potassium Hydride in Haloarene Reduction: CSNAr and Single Electron Transfer Reduction via Organic Electron Donors Formed in Benzene
[ASAP] Atom Efficient Synthesis of Selectively Difluorinated Carbocycles through a Gold(I)-Catalyzed Cyclization
A Practical and General Amidation Method from Isocyanates Enabled by Flow Technology
Arylfluorosulfate‐Based Electrophiles for Covalent Protein Labeling: A New Addition to the Arsenal

The potential of arylfluorosulfates for protein modification and the development of covalent inhibitors targeting lysine, tyrosine or serine are highlighted. The arylfluorosulfate group reacts slowly and enables high selectivity. Addition of arylfluorosulfates to the catalogue of available warheads promises exciting developments in chemical biology and drug discovery.
Abstract
Selective covalent modification of a targeted protein is a powerful tool in chemical biology and drug discovery, with applications ranging from identification and characterization of proteins and their functions to the development of targeted covalent inhibitors. Most covalent ligands contain an affinity motif and an electrophilic warhead that reacts with a nucleophilic residue of the targeted protein. Because the electrophilic warhead is prone to react and modify off‐target nucleophiles, its reactivity should be balanced carefully to maximize target selectivity. Arylfluorosulfates have recently emerged as latent electrophiles for selective labeling of context‐specific tyrosine and lysine residues in protein pockets. Here, we review the recent but intense introduction of arylfluorosulfates into the arsenal of available warheads for selective covalent modification of proteins. We highlight the untapped potential of this functional group for use in chemical biology and drug discovery.
[ASAP] Discovery of a Potent, Orally Bioavailable PI4KIIIß Inhibitor (UCB9608) Able To Significantly Prolong Allogeneic Organ Engraftment in Vivo
[ASAP] a-Chlorobenzylation of Nitroarenes via Vicarious Nucleophilic Substitution with Benzylidene Dichloride: Umpolung of the Friedel–Crafts Reaction
[ASAP] Regioselective Synthesis of Fluorosulfonyl 1,2,3-Triazoles from Bromovinylsulfonyl Fluoride
[ASAP] Potent and Highly Selective Inhibitors of the Proteasome Trypsin-like Site by Incorporation of Basic Side Chain Containing Amino Acid Derived Sulfonyl Fluorides
[3+2] Cycloaddition of an Azomethyne Ylide and Vinyl Sulfonyl Fluorides ― an Approach to Pyrrolidine‐3‐sulfonyl Fluorides
1-Bromoethene-1-sulfonyl fluoride (BESF) is another good connective hub for SuFEx click chemistry
DOI: 10.1039/C8CC03400A, Communication
1,2-Dibromoethane-1-sulfonyl fluoride (DESF): a bench stable and readily accessible precursor to BESF for the synthesis of [small beta]-sultams, sulfonyl fluoride substituted isoxazoles, triazoles and Michael adducts with SuFEx-able pendant sulfonyl fluoride handles.
The content of this RSS Feed (c) The Royal Society of Chemistry
[ASAP] Native Chemical Ligation–Photodesulfurization in Flow
[ASAP] Profiling and Application of Photoredox C(sp3)–C(sp2) Cross-Coupling in Medicinal Chemistry
[ASAP] Deoxyfluorination with Sulfonyl Fluorides: Navigating Reaction Space with Machine Learning
[ASAP] Discovery of Tetrahydroquinoxalines as Bromodomain and Extra-Terminal Domain (BET) Inhibitors with Selectivity for the Second Bromodomain
Cysteine-reactive probes and their use in chemical proteomics
DOI: 10.1039/C8CC01485J, Feature Article
In this Feature article, we provide an insight into different chemoproteomic probes and techniques to study cysteines in complex proteomes.
The content of this RSS Feed (c) The Royal Society of Chemistry
1-Bromoethene-1-sulfonyl fluoride (1-Br-ESF), a new SuFEx clickable reagent, and its application for regioselective construction of 5-sulfonylfluoro isoxazoles
DOI: 10.1039/C8CC00986D, Communication
A unique SuFEx clickable tris-electrophile, 1-Br-ESF, was developed and applied for the synthesis of 5-sulfonylfluoro isoxazoles with exclusive regioselectivity.
The content of this RSS Feed (c) The Royal Society of Chemistry
[ASAP] Benzoxazole-Linked Ultrastable Covalent Organic Frameworks for Photocatalysis
[ASAP] Small Molecule Interactome Mapping by Photoaffinity Labeling Reveals Binding Site Hotspots for the NSAIDs
[ASAP] Synthesis of Nitroolefins and Nitroarenes under Mild Conditions
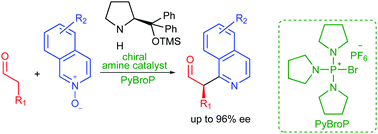
An organocatalytic enantioselective direct [small alpha]-heteroarylation of aldehydes with isoquinoline N-oxides
DOI: 10.1039/C8CC01735B, Communication
An enantioselective direct [small alpha]-heteroarylation of aldehydes with isoquinoline N-oxides has been realized. High enantiomeric excesses and moderate to good yields were achieved for a variety of [small alpha]-heteroarylated aldehydes.
The content of this RSS Feed (c) The Royal Society of Chemistry
Lewis acid catalyzed asymmetric [4+2] cycloaddition of cyclobutenones to synthesize [small alpha],[small beta]-unsaturated [small delta]-lactones
DOI: 10.1039/C8CC01040D, Communication
Here we report an efficient asymmetric cycloaddition of C[double bond, length as m-dash]O double bonds with cyclobutenones catalyzed by a chiral N,N[prime or minute]-dioxide/Yb(III) complex.
The content of this RSS Feed (c) The Royal Society of Chemistry
A New Portal to SuFEx Click Chemistry: A Stable Fluorosulfuryl Imidazolium Salt Emerging as an “F−SO2+” Donor of Unprecedented Reactivity, Selectivity, and Scope

A fluorosulfuryl imidazolium salt delivers an “F−SO2+” group to phenols and amines with phenomenal efficiency. In their Communication (DOI: 10.1002/anie.201712429), K. B. Sharpless, J. Dong, and co-workers present the practical, mole-scale synthesis of this stable, powerful reagent that provides clickable RNHSO2F, dramatically expands the scope of SuFEx products, and constitutes a complementary alternative to SO2F2 gas delivery.