31 May 05:06

Novel superacids: New Brønsted as well as Lewis superacids based on pentafluoroorthotellurates have been synthesized. The corresponding weakly coordinating anion has been used to stabilize carbocations, for example.
[Communication]
Anja Wiesner, Thomas W. Gries, Simon Steinhauer, Helmut Beckers, Sebastian Riedel
Angew. Chem. Int. Ed., May 30, 2017, https://doi.org/10.1002/anie.201702807 Read article
30 May 18:13
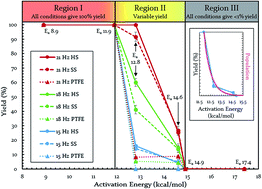
Chem. Sci., 2017, 8,5447-5453
DOI: 10.1039/C7SC00538E, Edge Article

Open Access
Joel M. Andersen, James Mack
We identified three different energetic regions that we believe are defining characteristics of most, if not all mechanochemical reactions. For a given ball mill's region, activation energy determines whether a reaction is energetically easy (Region I), challenging (Region II), or forbidden (Region III). In Region II, yield depends exponentially on oscillation frequency. Modifications granted control of the locations of Regions I, II, and III.
The content of this RSS Feed (c) The Royal Society of Chemistry

30 May 04:45

Integrative self-sorting of palladium(II) metal ions and banana-shaped ligands is driven by the interplay of complementary ligand geometries to form cis- or trans-[Pd2L2L′2] heteroleptic cage structures (1–3 in the picture). In their Communication (10.1002/anie.201702573), G. H. Clever and co-workers report a topologically unprecedented “doubly bridged figure eight” assembly in which one of the ligands is forced to adopt an anti-conformation. The extent of the geometric constraints in the heteroleptic cage system is further demonstrated through highly selective ligand-substitution reactions.
[Cover Picture]
Witold M. Bloch, Julian J. Holstein, Wolf Hiller, Guido H. Clever
Angew. Chem. Int. Ed., May 29, 2017, https://doi.org/10.1002/anie.201704764 Read article
27 May 09:42
by Bahiru Punja Benke, Pulakesh Aich, Younghoon Kim, Kyung Lock Kim, Md Rumum Rohman, Soonsang Hong, In-Chul Hwang, Eun Hui Lee, Joon Ho Roh and Kimoon Kim

Journal of the American Chemical Society
DOI: 10.1021/jacs.7b02708

25 May 20:12
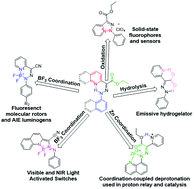
Chem. Commun., 2017, 53,6674-6684
DOI: 10.1039/C7CC02879B, Feature Article
Ivan Aprahamian
This feature article surveys the various ways by which a structurally simple hydrazone can be used in accessing different functional materials, mainly photo/chemically activated switches, fluorophores and sensors.
The content of this RSS Feed (c) The Royal Society of Chemistry

23 May 13:40
by Jack Chen, Subhabrata Maiti, Ilaria Fortunati, Camilla Ferrante, Leonard J. Prins
Abstract
The next generation of adaptive, intelligent chemical systems will rely on a continuous supply of energy to maintain the functional state. Such systems will require chemical methodology that provides precise control over the energy dissipation process, and thus, the lifetime of the transiently activated function. This manuscript reports on the use of structurally diverse chemical fuels to control the lifetime of two different systems under dissipative conditions: transient signal generation and the transient formation of self-assembled aggregates. The energy stored in the fuels is dissipated at different rates by an enzyme, which installs a dependence of the lifetime of the active system on the chemical structure of the fuel. In the case of transient signal generation, it is shown that different chemical fuels can be used to generate a vast range of signal profiles, allowing temporal control over two orders of magnitude. Regarding self-assembly under dissipative conditions, the ability to control the lifetime using different fuels turns out to be particularly important as stable aggregates are formed only at well-defined surfactant/fuel ratios, meaning that temporal control cannot be achieved by simply changing the fuel concentration.

Chemical fuels to control lifetime: The next generation of adaptive, intelligent materials will require precise control over energy dissipation processes. Towards this goal, structurally diverse chemical fuels are used to control the lifetime of two such processes: transient signal generation and transient self-assembly.
21 May 16:44
by Andrew I. Cooper

ACS Central Science
DOI: 10.1021/acscentsci.7b00146
17 May 19:37

A new type of twisted nanographene containing an [8]circulene moiety in a polycyclic framework of 96 sp2 carbon atoms was synthesized from a macrocyclic diyne (see picture). Its structure was studied with X-ray crystallography and DFT calculations.
[Communication]
Kwan Yin Cheung, Chi Kit Chan, Zhifeng Liu, Qian Miao
Angew. Chem. Int. Ed., May 16, 2017, https://doi.org/10.1002/anie.201703754 Read article
16 May 19:46
by Anindita Das, Ghislaine Vantomme, Albert J. Markvoort, Huub M. M. ten Eikelder, Miguel Garcia-Iglesias, Anja R. A. Palmans and E. W. Meijer

Journal of the American Chemical Society
DOI: 10.1021/jacs.7b02835

16 May 04:46
by Abdel Monem Rawashdeh, Priyakumari Chakkingal Parambil, Tao Zeng and Roald Hoffmann

Journal of the American Chemical Society
DOI: 10.1021/jacs.7b03388

16 May 04:43

A new resist material for electron beam lithography has been created that is based on a supramolecular assembly. Based on this supramolecular approach, high-resolution structures can be written that show unprecedented selectivity when exposed to etching conditions involving plasmas.
[Communication]
Scott M. Lewis, Antonio Fernandez, Guy A. DeRose, Matthew S. Hunt, George F. S. Whitehead, Agnese Lagzda, Hayden R. Alty, Jesus Ferrando-Soria, Sarah Varey, Andreas K. Kostopoulos, Fredrik Schedin, Christopher A. Muryn, Grigore A. Timco, Axel Scherer, Stephen G. Yeates, Richard E. P. Winpenny
Angew. Chem. Int. Ed., May 15, 2017, https://doi.org/10.1002/anie.201700224 Read article
09 May 04:33
by Dawei Zhang, Tanya K. Ronson, Jesús Mosquera, Alexandre Martinez, Laure Guy and Jonathan R. Nitschke

Journal of the American Chemical Society
DOI: 10.1021/jacs.7b02950

06 May 07:41

Spark discharge in a deuterium world: Repeating the iconic Miller–Urey experiment in a fully deuterated environment produces a different product distribution to that found in the non-deuterated control, giving insights into how isotopic enrichment and processing may lead to significant additional complexity in chemical space under chemically identical reaction conditions.
[Communication]
Geoffrey J. T. Cooper, Andrew J. Surman, Jim McIver, Stephanie M. Colón-Santos, Piotr S. Gromski, Saskia Buchwald, Irene Suárez Marina, Leroy Cronin
Angew. Chem. Int. Ed., May 05, 2017, https://doi.org/10.1002/anie.201610837 Read article
05 May 07:44

Protein ions in extremely high charge states are sufficiently acidic that noble gases can be protonated in ion–molecule reactions. The results are not consistent with the charge residue model for the formation of highly charged protein ions in electrospray ionization (ESI) because the protein ions are more than 375 kJ mol−1 less basic than the least volatile components of ESI solutions. The chain ejection model is more likely.
[Communication]
Muhammad A. Zenaidee, Michael G. Leeming, Fangtong Zhang, Toby T. Funston, William A. Donald
Angew. Chem. Int. Ed., May 04, 2017, https://doi.org/10.1002/anie.201702781 Read article
04 May 20:09
by Sarah J. Pike, Jordan J. Hutchinson and Christopher A. Hunter

Journal of the American Chemical Society
DOI: 10.1021/jacs.7b02008

04 May 04:29

If at first you don't succeed, try, try again: With enzymes not available on the early Earth, energy-dissipative cycles might have contributed to the correction and recycling of ribonucleic acids, which defines a plausible scenario for the prebiotic evolution of RNA.
[Communication]
Angelica Mariani, John D. Sutherland
Angew. Chem. Int. Ed., May 03, 2017, https://doi.org/10.1002/anie.201703169 Read article
03 May 17:23

Unbreakable polymers: Random covalent intramolecular cross-links in single-chain polymer nanoparticles break preferably during ultrasonication, making the polymers resistant to mechanochemistry in solution. Above a certain cross-link density, the nanoparticles become virtually invincible to shear in solution.
[Communication]
Avishai Levy, Feng Wang, Arad Lang, Or Galant, Charles E. Diesendruck
Angew. Chem. Int. Ed., May 02, 2017, https://doi.org/10.1002/anie.201612242 Read article
30 Apr 21:51

Weighty evidence: Molecular torsion balances containing an N-aryl imide and an additional aromatic moiety provided direct experimental evidence for halogen–aryl π interactions in solution (see picture). The magnitude of the halogen–aryl π interactions in the unimolecular systems described herein are found to be larger than −5.0 kJ mol−1.
[Communication]
Han Sun, André Horatscheck, Vera Martos, Max Bartetzko, Ulrike Uhrig, Dieter Lentz, Peter Schmieder, Marc Nazaré
Angew. Chem. Int. Ed., April 28, 2017, https://doi.org/10.1002/anie.201700520 Read article
28 Apr 13:44
New J. Chem., 2017, 41,4262-4268
DOI: 10.1039/C7NJ00182G, Focus
Alexander Yu. Rulev
The phenomenon of serendipitous discoveries in chemistry has been discussed.
The content of this RSS Feed (c) The Royal Society of Chemistry

28 Apr 04:52
by Carlo Bravin, Elena Badetti, Francesca A. Scaramuzzo, Giulia Licini and Cristiano Zonta

Journal of the American Chemical Society
DOI: 10.1021/jacs.7b02341

27 Apr 18:50
by Kim, H., Yang, S., Rao, S. R., Narayanan, S., Kapustin, E. A., Furukawa, H., Umans, A. S., Yaghi, O. M., Wang, E. N.
Atmospheric water is a resource equivalent to ~10% of all fresh water in lakes on Earth. However, an efficient process for capturing and delivering water from air, especially at low humidity levels (down to 20%), has not been developed. We report the design and demonstration of a device based on a porous metal-organic framework {MOF-801, [Zr6O4(OH)4(fumarate)6]} that captures water from the atmosphere at ambient conditions by using low-grade heat from natural sunlight at a flux of less than 1 sun (1 kilowatt per square meter). This device is capable of harvesting 2.8 liters of water per kilogram of MOF daily at relative humidity levels as low as 20% and requires no additional input of energy.
25 Apr 20:05
by Li Yu, Jian Sun, Qian Wang, Yan Guan, Le Zhou, Jingxuan Zhang, Lanying Zhang, Huai Yang
Controllable assembly of molecular motors on solid surfaces is a fundamental issue for providing them to perform physical tasks. However, it can hardly be achieved by most previous methods due to their inherent limitations. Here, a general strategy is designed for the reprogrammable assembly of molecular motors on solid surfaces based on dynamic bonds. In this method, molecular motors with disulfide bonds can be remotely, reversibly, and precisely attached to solid surfaces with disulfide bonds, regardless of their chemical composition and microstructure. More importantly, it not only allows encoding geometric information referring to a pattern of molecular motors, but also enables erasing and re-encoding of geometric information via hemolytic photocleavage and recombination of disulfide bonds. Thus, solid surfaces can be regarded as “computer hardware”, where molecular motors can be reformatted and reprogramed as geometric information.

Dynamic disulfide bonds are introduced into the chemical structure of molecular motors and the surface of solid substrates so that molecular motors can reversibly and selectively assemble on solid surfaces regardless of their chemical compositions and microstructures, while maintaining photochemistry isomerization behavior. Due to the reversible photoinduced exchange reaction, molecular motors can be reformatted and reprogramed repeatedly on solid surfaces.
25 Apr 20:04
by Martin Ferus, Fabio Pietrucci, Antonino Marco Saitta, Antonin Knižek, Petr Kubelik, Ondřeȷ Ivanek, Violetta Shestivska, Svatopluk Civiš
The Miller–Urey experiments pioneered modern research on the molecular origins of life, but their actual relevance in this field was later questioned because the gas mixture used in their research is considered too reducing with respect to the most accepted hypotheses for the conditions on primordial Earth. In particular, the...
21 Apr 21:00
by Zhang, R., Murata, M., Wakamiya, A., Shimoaka, T., Hasegawa, T., Murata, Y.
Dissociation of an acid molecule in aqueous media is one of the most fundamental solvation processes but its details remain poorly understood at the distinct molecular level. Conducting high-pressure treatments of an open-cage fullerene C70 derivative with hydrogen fluoride (HF) in the presence of H2O, we achieved an unprecedented encapsulation of H2O·HF and H2O. Restoration of the opening yielded the endohedral C70s, that is, (H2O·HF)@C70, H2O@C70, and HF@C70 in macroscopic scales. Putting an H2O·HF complex into the fullerene cage was a crucial step, and it would proceed by the synergistic effects of "pushing from outside" and "pulling from inside." The structure of the H2O·HF was unambiguously determined by single crystal x-ray diffraction analysis. The nuclear magnetic resonance measurements revealed the formation of a hydrogen bond between the H2O and HF molecules without proton transfer even at 140°C.
21 Apr 04:27
by Yoshihiro Ueda, Hiroaki Ito, Daishi Fujita and Makoto Fujita

Journal of the American Chemical Society
DOI: 10.1021/jacs.7b02745

16 Apr 12:04
by Choi, J., Fu, G. C.
Because the backbone of most organic molecules is composed primarily of carbon-carbon bonds, the development of efficient methods for their construction is one of the central challenges of organic synthesis. Transition metal–catalyzed cross-coupling reactions between organic electrophiles and nucleophiles serve as particularly powerful tools for achieving carbon-carbon bond formation. Until recently, the vast majority of cross-coupling processes had used either aryl or alkenyl electrophiles as one of the coupling partners. In the past 15 years, versatile new methods have been developed that effect cross-couplings of an array of alkyl electrophiles, thereby greatly expanding the diversity of target molecules that are readily accessible. The ability to couple alkyl electrophiles opens the door to a stereochemical dimension—specifically, enantioconvergent couplings of racemic electrophiles—that substantially enhances the already remarkable utility of cross-coupling processes.
13 Apr 22:22
by Guillaume Povie
The synthesis of a carbon nanobelt, comprising a closed loop of fully fused edge-sharing benzene rings, has been an elusive goal in organic chemistry for more than 60 years. Here we report the synthesis of one such compound through iterative Wittig reactions followed by a nickel-mediated aryl-aryl coupling reaction. The cylindrical shape of its belt structure was confirmed by x-ray crystallography, and its fundamental optoelectronic properties were elucidated by ultraviolet-visible absorption, fluorescence, and Raman spectroscopic studies, as well as theoretical calculations. This molecule could potentially serve as a seed for the preparation of structurally well-defined carbon nanotubes.
Authors: Guillaume Povie, Yasutomo Segawa, Taishi Nishihara, Yuhei Miyauchi, Kenichiro Itami
13 Apr 22:18
by Junwon Choi
Because the backbone of most organic molecules is composed primarily of carbon-carbon bonds, the development of efficient methods for their construction is one of the central challenges of organic synthesis. Transition metal–catalyzed cross-coupling reactions between organic electrophiles and nucleophiles serve as particularly powerful tools for achieving carbon-carbon bond formation. Until recently, the vast majority of cross-coupling processes had used either aryl or alkenyl electrophiles as one of the coupling partners. In the past 15 years, versatile new methods have been developed that effect cross-couplings of an array of alkyl electrophiles, thereby greatly expanding the diversity of target molecules that are readily accessible. The ability to couple alkyl electrophiles opens the door to a stereochemical dimension—specifically, enantioconvergent couplings of racemic electrophiles—that substantially enhances the already remarkable utility of cross-coupling processes.
Authors: Junwon Choi, Gregory C. Fu
13 Apr 12:03
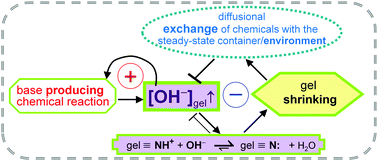
Chem. Commun., 2017, 53,4973-4976
DOI: 10.1039/C7CC02497E, Communication
J. Horvath
Periodic length changes, over 20%, were sustained in a pH-responsive gel by associating the methylene glycol-sulphite OH-producing clock-reaction with variations of the exchange time induced between the core of the gel and a steady chemical environment.
The content of this RSS Feed (c) The Royal Society of Chemistry

13 Apr 04:28
by Sambasiva R. Bheemireddy, Matthew P. Hautzinger, Tao Li, Byeongdu Lee and Kyle N. Plunkett

Journal of the American Chemical Society
DOI: 10.1021/jacs.6b12916


 Open Access
Open Access