Shared posts
Diversity-oriented heterocyclic synthesis using divergent reactivity of N-substituted iso(thio)cyanates
DOI: 10.1039/C5CC07212C, Communication
Reactions involving N-substituted isocyanates and isothiocyanates are compared: divergent reactivity is observed in cascade reactions.
The content of this RSS Feed (c) The Royal Society of Chemistry
One-Pot Synthesis of Aza-Diketopiperazines Enabled by Controlled Reactivity of N-Isocyanate Precursors
Cascade reactions of nitrogen-substituted isocyanates: a new tool in heterocyclic chemistry
DOI: 10.1039/C5SC03197D, Edge Article
 Open Access
Open Access   This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
  This article is licensed under a Creative Commons Attribution 3.0 Unported Licence.
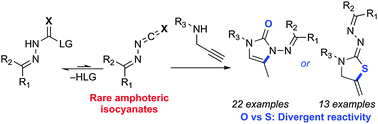
Rare and amphoteric intermediates, in situ generation: controlled reactivity in diverse cascade reactions, over 100 examples.
To cite this article before page numbers are assigned, use the DOI form of citation above.
The content of this RSS Feed (c) The Royal Society of Chemistry
UCLA Professor Patrick Harran Strikes Deal with Prosecutors
 Patrick Harran, the UCLA professor who faced four felony counts in connection with the death of Sheri Sangji in a laboratory fire, has struck a deal with prosecutors that allows him to avoid charges in exchange for a $10,000 fine, 800 hours of community service, and running a lab free of safety violations. So long as Harran completes his end of the terms of the agreement, he will avoid trial and have an untarnished criminal record.
Patrick Harran, the UCLA professor who faced four felony counts in connection with the death of Sheri Sangji in a laboratory fire, has struck a deal with prosecutors that allows him to avoid charges in exchange for a $10,000 fine, 800 hours of community service, and running a lab free of safety violations. So long as Harran completes his end of the terms of the agreement, he will avoid trial and have an untarnished criminal record.
What a relief!
As an assistant professor in charge of a research lab, I could not be happier with this outcome. I have a lot of stuff to worry about, and ensuring the safety of my students cannot be allowed to get in the way of important things like finding consulting gigs, collecting awards, traveling to international conferences, and stealing ideas for grants. All of those OSHA rules are meant for industry, not academia. The bar for what passes as safe in academic labs is clear, and people who want to work under moderately safe conditions know better than to go to graduate school. The government simply can’t expect me to be responsible for what happens in my lab, which is well over 50 feet from my office and not even the same direction as the restroom. I’m happy to buy enough safety goggles and almost enough lab coats to outfit my students, but the rest is up to them. If Aldrich has written a technical note on their hazardous experiment, my students know not to bother me.
The most important aspect of the Harran deal is how it extends the long, proud tradition of excusing PIs of any professional responsibility for their work. Society recognizes that professors are only supposed to have good things happen to them. We get the lion’s share of credit for papers, not the students or postdocs. We get the big salaries, not the students or postdocs. We get the awards, not the students or postdocs. On the flip side, professors must be protected from negative consequences at all costs. If an accident happens in one of our labs, that’s the students’ fault. If multiple papers from one of our labs contain fabricated data, that’s the students’ fault as well. Clearly, professors are not responsible for supervising their groups for integrity or safety. We know this because Dalibor Sames and Patrick Harran are still in charge of their labs. I applaud Columbia and UCLA for recognizing that you can’t discriminate against professors for trivial things like irresponsibility and incompetence. Anyway, it’s the competent professors you need to watch—lightning never strikes twice, right?
Of course, I realize that there should be some consequences when something truly horrible happens. In these situations, professors must arrange for perfunctory punishments that allow all of the parties charged with oversight to save face. That’s what we saw here: UCLA threw some money at a scholarship in the victim’s name and at establishing a safety program it should have had in the first place. Personally, Harran was forced to donate money to the hospital where his student died. Incidentally, I think $10,000 was way too much; the man only earns $301,000 a year. How is he going to make ends meet with just $291,000? At least Harran’s lawyers were clever in how they disguised the 800 hours of community service as a major inconvenience instead of court-mandated preparation for the Broader Impacts section of Harran’s next NSF proposal. Killing two birds with one stone is exactly why good lawyers get paid the big bucks.
—
In all seriousness, I think the deal agreed to by prosecutors is a grave injustice, but one that comes as no surprise in today’s legal system. Without any changes to the material facts of the case, how does the DA go from charging someone with four felony counts to striking a deal that allows Harran to have a spotless record with a payment, community service, and actually doing his job of running a lab free of safety violations? Note that this was not a plea bargain; Harran pleaded guilty to nothing—not a misdemeanor, or even an infraction.
The game plan of Harran’s legal defense was quite effective: delay, delay, and delay. They gummed up the works with continuance after continuance and motion after motion. In the end, it appeared the prosecutors were willing to do anything just to clear the case. I mean, was this deal what the prosecutors were holding out for all of these years? What makes it all the more worse is that the original deal called for 400 rather than 800 hours of community service. The judge had to step in and double it.
My heart goes out to Sheri’s family for their loss. While I think our legal system has denied them justice, my hope is that the field of chemistry does not forget what happened to her. I hope UCLA’s reported new-and-improved safety culture persists, and I hope the rest of the world of academic chemistry also strives to do a much better job regarding safety than it has in the past. At the very least, I can guarantee you that Sheri’s death has had an indelible, positive effect my approach to safety and how I manage my lab and students.
For more coverage: C&EN’s Jyllian Kemsley and Michael Torrice have done a fantastic service for the community in covering the case, and Chemjobber has been curating links to coverage on his site.
Studies on Difficult Intramolecular Hydroaminations in the Context of Four Syntheses of Alkaloid Natural Products
Diversity-Oriented Synthesis of Hydrazine-Derived Compounds from Amino Isocyanates Generated In Situ
Jean-François V-RNice paper :)

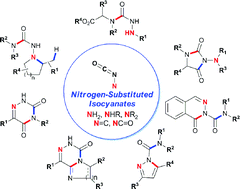
Behind the mask: Nitrogen-substituted isocyanates are rare and their synthetic potential is virtually untapped. Simple masked precursors can form amphoteric amino isocyanate intermediates in situ, and allows the synthesis of complex hydrazine derivatives upon addition with amines. This reactivity was used in a cascade substitution/hydroamination sequence, and in the assembly of azadipeptide analogues.
Low-Temperature Rh-Catalyzed Asymmetric 1,4-Addition of Arylboronic Acids to α,β-Unsaturated Carbonyl Compounds
A Practical Access to Highly Enantiomerically Pure Flavanones by Catalytic Asymmetric Transfer Hydrogenation
Jean-François V-RAmanda might be good for you
Synthesis of Azomethine Imines Using an Intramolecular Alkyne Hydrohydrazination Approach
Selective Intramolecular Palladium(II)-Catalyzed Aminooxygenation vs. Diamination of Alkenylureas: Efficient Microwave-Assisted Reactions to Bicyclic Piperazinones
Jean-François V-RNico tu pourrais cite ca pour ton papier
Abstract
Alkenylureas arising from glycine allylamides were proven to be suitable substrates for the synthesis of bicyclic five-membered ring-fused piperazinones. The reported intramolecular domino processes, performed under oxidative conditions with bis(acetonitrile)palladium dichloride as catalyst and copper(II) chloride in a stoichiometric amount by microwave activation, were completely selective, involving either diamination or aminooxygenation. While the latter process is determined by the direct intervention of the urea oxygen on the σ-alkylpalladium intermediate, the diamination reaction can in principle derive from a direct attack of the second nitrogen atom on the palladium complex or on the first formed chloromethylpiperazinone. Indeed, this latter species was isolated and proved to be capable of conversion solely into the imidazopiperazinone.

Copper-catalyzed asymmetric 1,4-conjugate addition of Grignard reagents to linear [small alpha],[small beta],[gamma],[small delta]-unsaturated ketones
DOI: 10.1039/C3CC42088D, Communication
A highly regioselective and enantioselective copper-catalyzed 1,4-conjugate addition of Grignard reagents to linear [small alpha],[small beta],[gamma],[small delta]-unsaturated ketones was developed.
The content of this RSS Feed (c) The Royal Society of Chemistry