24 Oct 20:19
Chem. Commun., 2016, 52,13675-13678
DOI: 10.1039/C6CC08113D, Communication
Bo Qiao, Yun Liu, Semin Lee, Maren Pink, Amar H. Flood
We show a high-yielding synthesis (80%) of [3]rotaxanes using shape-persistent macrocycles, cyanostars, and anionic organo-phosphate templates catalyzed by low steric-demand click chemistry.
The content of this RSS Feed (c) The Royal Society of Chemistry

19 Oct 09:18
by Semin Lee, Etienne Chénard, Danielle L. Gray and Jeffrey S. Moore

Journal of the American Chemical Society
DOI: 10.1021/jacs.6b08752

16 Oct 14:13

Space, finally! The explicit investigation of perylenediimides for anion–π catalysis reveals a remarkably rich picture dominated by giant quadrupole moments, space to accommodate larger transformations, highest enantioselectivities so far, and decreasing activity with increasing twist.
[Communication]
Chao Wang, François N. Miros, Jiri Mareda, Naomi Sakai, Stefan Matile
Angew. Chem. Int. Ed., October 14, 2016, DOI: 10.1002/anie.201608842. Read article
10 Oct 18:03
by Hongwei Zhou

Nature Chemistry.
doi:10.1038/nchem.2625
Authors: Hongwei Zhou, Changguo Xue, Philipp Weis, Yasuhito Suzuki, Shilin Huang, Kaloian Koynov, Günter K. Auernhammer, Rüdiger Berger, Hans-Jürgen Butt & Si Wu
Reversibly inducing solid-to-liquid transitions of polymers at room temperature represents a challenge for enhanced processability and applications of polymers. Now, three azopolymers have been shown to exhibit photoswitchable glass transition temperatures, resulting in reversible, solid-to-liquid transitions. Light exposure can heal cracks in hard azopolymers, reduce surface roughness of azopolymer films and control azopolymer adhesion.
10 Oct 05:55
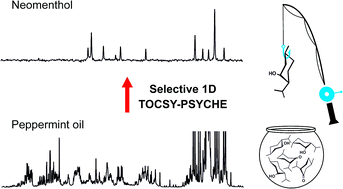
RSC Adv., 2016, 6,100063-100066
DOI: 10.1039/C6RA22807K, Communication
G. Dal Poggetto, L. Castanar, G. A. Morris, M. Nilsson
A new NMR experiment aids the identification of components in complex systems, including mixtures.
The content of this RSS Feed (c) The Royal Society of Chemistry

08 Oct 08:20
![[N⋅⋅⋅I+⋅⋅⋅N] Halogen‐Bonded Dimeric Capsules from Tetrakis(3‐pyridyl)ethylene Cavitands [N⋅⋅⋅I+⋅⋅⋅N] Halogen‐Bonded Dimeric Capsules from Tetrakis(3‐pyridyl)ethylene Cavitands](http://onlinelibrary.wiley.com/store/10.1002/anie.201607789/asset/image_n/anie201607789-toc-0001.png?v=1&s=6bf29ba005fb6f0b2857586b3ca872d9f1fdad8b)
I move in: A tetrakis(3-pyridyl)ethylene cavitand reacts with silver tosylate forming a dimeric capsule through four [N⋅⋅⋅Ag+⋅⋅⋅N] coordination bonds. Treatment of this Ag-capsule with I2 leads, via precipitation of AgI, to the corresponding [N⋅⋅⋅I+⋅⋅⋅N] halogen-bonded capsule.
[Communication]
Lotta Turunen, Ulrike Warzok, Rakesh Puttreddy, Ngong Kodiah Beyeh, Christoph A. Schalley, Kari Rissanen
Angew. Chem. Int. Ed., October 06, 2016, DOI: 10.1002/anie.201607789. Read article
08 Oct 08:19
Marcin A. Majewski, Yongseok Hong, Prof. Tadeusz Lis, Dr. Janusz Gregoliński, Prof. Piotr J. Chmielewski, Dr. Joanna Cybińska, Prof. Dongho Kim and Prof. Marcin Stępień

A non-Euclidean belt: Octulene, the largest coronoid ring reported to date, features a saddle-shaped aromatic hydrocarbon ring that fits on a hyperbolic surface. The large central cavity, in spite of its low electrostatic potential, acts as an anion receptor in nonpolar media, with a particularly good affinity for the chloride anion.
Read more now
06 Oct 17:24
by Witold M. Bloch, Yoko Abe, Julian J. Holstein, Claudia M. Wandtke, Birger Dittrich and Guido H. Clever

Journal of the American Chemical Society
DOI: 10.1021/jacs.6b08694

05 Oct 17:42
by Ortega-Arroyo, J., Bissette, A. J., Kukura, P., Fletcher, S. P.
Autocatalytic chemical reactions are widely studied as models of biological processes and to better understand the origins of life on Earth. Minimal self-reproducing amphiphiles have been developed in this context and as an approach to de novo “bottom–up” synthetic protocells. How chemicals come together to produce living systems, however, remains...
05 Oct 04:18

A chemical reaction network for small molecules is reported that provides a rapid adaptation response to out-of-equilibrium conditions inside an evaporating droplet. The chemical network provides control over the molecular constitution and spatial arrangement of the deposited solute.
[Communication]
Joseph J. Armao, Jean-Marie Lehn
Angew. Chem. Int. Ed., October 04, 2016, DOI: 10.1002/anie.201606546. Read article
04 Oct 21:04
by Philipp J. Altmann and Alexander Pöthig

Journal of the American Chemical Society
DOI: 10.1021/jacs.6b08571

04 Oct 18:04
Chem. Commun., 2016, 52,12669-12671
DOI: 10.1039/C6CC06716F, Communication
Xuan-Xuan Chen, Yun-Bao Jiang, Eric V. Anslyn
A supramolecular polymeric system that shows an unusual "racemate-rules" chiroptical property, an effect opposite to the well-known "majority-rules", has been utilized for accurate determination of malic acid enantiopurity at high ee values.
The content of this RSS Feed (c) The Royal Society of Chemistry

03 Oct 20:30
by Yan Qiao

Nature Chemistry.
doi:10.1038/nchem.2617
Authors: Yan Qiao, Mei Li, Richard Booth & Stephen Mann
The chemical construction of compartmentalized colloidal objects with biomimetic functions and collective properties is a key challenge in synthetic protocell research. Now, an interacting binary community of protocells has been designed to display artificial predatory behaviour in which protease-containing coacervate microdroplets obliterate a population of proteinosomes, and capture a chemical payload via a simple trafficking process.
30 Sep 07:43
by Guzmán Gil-Ramírez, Steven Hoekman, Matthew O. Kitching, David A. Leigh, Iñigo J. Vitorica-Yrezabal and Gen Zhang

Journal of the American Chemical Society
DOI: 10.1021/jacs.6b08421

28 Sep 21:00
by Sergey N. Semenov
Autocatalytic, bistable, oscillatory networks of biologically relevant organic reactions
Nature 537, 7622 (2016). doi:10.1038/nature19776
Authors: Sergey N. Semenov, Lewis J. Kraft, Alar Ainla, Mengxia Zhao, Mostafa Baghbanzadeh, Victoria E. Campbell, Kyungtae Kang, Jerome M. Fox & George M. Whitesides
Networks of organic chemical reactions are important in life and probably played a central part in its origin. Network dynamics regulate cell division, circadian rhythms, nerve impulses and chemotaxis, and guide the development of organisms. Although out-of-equilibrium networks of chemical reactions have the potential to display emergent network dynamics such as spontaneous pattern formation, bistability and periodic oscillations, the principles that enable networks of organic reactions to develop complex behaviours are incompletely understood. Here we describe a network of biologically relevant organic reactions (amide formation, thiolate–thioester exchange, thiolate–disulfide interchange and conjugate addition) that displays bistability and oscillations in the concentrations of organic thiols and amides. Oscillations arise from the interaction between three subcomponents of the network: an autocatalytic cycle that generates thiols and amides from thioesters and dialkyl disulfides; a trigger that controls autocatalytic growth; and inhibitory processes that remove activating thiol species that are produced during the autocatalytic cycle. In contrast to previous studies that have demonstrated oscillations and bistability using highly evolved biomolecules (enzymes and DNA) or inorganic molecules of questionable biochemical relevance (for example, those used in Belousov–Zhabotinskii-type reactions), the organic molecules we use are relevant to metabolism and similar to those that might have existed on the early Earth. By using small organic molecules to build a network of organic reactions with autocatalytic, bistable and oscillatory behaviour, we identify principles that explain the ways in which dynamic networks relevant to life could have developed. Modifications of this network will clarify the influence of molecular structure on the dynamics of reaction networks, and may enable the design of biomimetic networks and of synthetic self-regulating and evolving chemical systems.

28 Sep 05:18

The spin within: A carefully designed triarylphosphine oxide scaffold with sterically demanding spirofluorenyl moieties undergoes chemical one-electron reduction at its phosphoryl moiety. The unique stability of the formed radical anion enables the isolation and X-ray crystallographic characterization of this hitherto elusive species.
[Communication]
Tobias A. Schaub, Eva M. Zolnhofer, Dominik P. Halter, Tatyana E. Shubina, Frank Hampel, Karsten Meyer, Milan Kivala
Angew. Chem. Int. Ed., September 27, 2016, DOI: 10.1002/anie.201605963. Read article
26 Sep 19:26
by Melissa Ball, Yu Zhong, Brandon Fowler, Boyuan Zhang, Panpan Li, Grisha Etkin, Daniel W. Paley, John Decatur, Ankur K. Dalsania, Hexing Li, Shengxiong Xiao, Fay Ng, Michael L. Steigerwald and Colin Nuckolls

Journal of the American Chemical Society
DOI: 10.1021/jacs.6b05474

26 Sep 19:13
by Takuya Ishii, Katsunori Suzuki, Taichi Nakamura and Makoto Yamashita

Journal of the American Chemical Society
DOI: 10.1021/jacs.6b08714

24 Sep 20:40
by Dalal Alezi, Ioannis Spanopoulos, Constantinos Tsangarakis, Aleksander Shkurenko, Karim Adil, Youssef Belmabkhout, Michael O′Keeffe, Mohamed Eddaoudi and Pantelis N. Trikalitis

Journal of the American Chemical Society
DOI: 10.1021/jacs.6b08176

22 Sep 11:19
Chem. Commun., 2016, 52,12322-12325
DOI: 10.1039/C6CC06842A, Communication
Monica I. Rednic, Richard A. Varga, Attila Bende, Ioana G. Grosu, Maria Miclaus, Niculina D. Hadade, Anamaria Terec, Elena Bogdan, Ion Grosu
Exciting [small beta]-HCH-anion recognition via cooperative aliphatic C-H-anion contacts is discussed.
The content of this RSS Feed (c) The Royal Society of Chemistry

19 Sep 19:59
by Jia-Cheng Chang

Nature Chemistry.
doi:10.1038/nchem.2608
Authors: Jia-Cheng Chang, Shin-Han Tseng, Chien-Chen Lai, Yi-Hung Liu, Shie-Ming Peng & Sheng-Hsien Chiu
By exploiting structural rigidity, coordination geometries and bond rotational barriers that disfavour the formation of smaller homologues, molecular switches based on [c3] and [c4]daisy chains have been assembled selectively; they display muscle-like motion in multiple dimensions with changes in length of approximately 23% and 36%, respectively.
17 Sep 10:57

Should I stay or should I go? Static and dynamic sheets are formed from selective self-assembly of geometric macrocycle isomers based on anthracene units. The self-assembly of the cis isomer generates static planar sheets (left), whereas the trans isomer forms dynamic rolled sheets (right), which are reversibly unrolled upon stimulation by a thermal signal.
[Communication]
Yanqiu Wang, Yongju Kim, Myongsoo Lee
Angew. Chem. Int. Ed., September 16, 2016, DOI: 10.1002/anie.201607143. Read article
17 Sep 10:57

Switching on catalysis: A new class of synthetic receptors exhibit enzyme-like properties in organic solvents. Their catalytic activity (see picture; green arrow) can be turned on by a size and shape match of the substrate with the binding pocket. The electrostatic environment of the active site can be further modulated using cationic regulators (blue arrow).
[Communication]
Yupu Qiao, Long Zhang, Jia Li, Wei Lin, Zhenqiang Wang
Angew. Chem. Int. Ed., September 16, 2016, DOI: 10.1002/anie.201606847. Read article
14 Sep 17:51
by Qi Xiao, Jack D. Rubien, Zhichun Wang, Ellen H. Reed, Daniel A. Hammer, Dipankar Sahoo, Paul A. Heiney, Srujana S. Yadavalli, Mark Goulian, Samantha E. Wilner, Tobias Baumgart, Sergei A. Vinogradov, Michael L. Klein and Virgil Percec

Journal of the American Chemical Society
DOI: 10.1021/jacs.6b08069

14 Sep 17:50
by Ting-Zheng Xie, Kevin J. Endres, Zaihong Guo, James M. Ludlow, Charles N. Moorefield, Mary Jane Saunders, Chrys Wesdemiotis and George R. Newkome

Journal of the American Chemical Society
DOI: 10.1021/jacs.6b07969

14 Sep 04:17
by Nicolas Kaeffer, Julien Massin, Colette Lebrun, Olivier Renault, Murielle Chavarot-Kerlidou and Vincent Artero

Journal of the American Chemical Society
DOI: 10.1021/jacs.6b05865

14 Sep 04:17
by Jonathan J. Danon, David A. Leigh, Paul R. McGonigal, John W. Ward and Jhenyi Wu

Journal of the American Chemical Society
DOI: 10.1021/jacs.6b07733

12 Sep 14:26
Chem. Commun., 2016, 52,11783-11786
DOI: 10.1039/C6CC06059E, Communication
Simon Donck, Jennifer Fize, Edmond Gravel, Eric Doris, Vincent Artero
A carbon nanotube-cobaloxime H2-evolving nanohybrid catalyst was prepared through supramolecular assembly of tailored polymerizable amphiphiles, leading to the coordination of cobalt on pyridine-coated nanotubes.
The content of this RSS Feed (c) The Royal Society of Chemistry

12 Sep 13:12
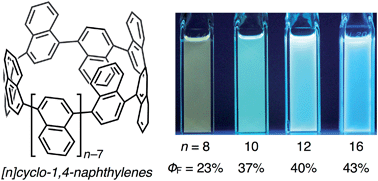
Chem. Sci., 2016, Advance Article
DOI: 10.1039/C6SC04048A, Edge Article

Open Access
Keishu Okada, Akiko Yagi, Yasutomo Segawa, Kenichiro Itami
The synthesis and properties of various [n]cyclo-1,4-naphthylenes ([n]CNs, n = 8, 10, 12, and 16) are described.
To cite this article before page numbers are assigned, use the DOI form of citation above.
The content of this RSS Feed (c) The Royal Society of Chemistry

06 Sep 19:52
by Hai-Sen Xu, San-Yuan Ding, Wan-Kai An, Han Wu and Wei Wang

Journal of the American Chemical Society
DOI: 10.1021/jacs.6b07516





![[N⋅⋅⋅I+⋅⋅⋅N] Halogen‐Bonded Dimeric Capsules from Tetrakis(3‐pyridyl)ethylene Cavitands [N⋅⋅⋅I+⋅⋅⋅N] Halogen‐Bonded Dimeric Capsules from Tetrakis(3‐pyridyl)ethylene Cavitands](http://onlinelibrary.wiley.com/store/10.1002/anie.201607789/asset/image_n/anie201607789-toc-0001.png?v=1&s=6bf29ba005fb6f0b2857586b3ca872d9f1fdad8b)




















 Open Access
Open Access
