Shared posts
Structural Investigation of Graphitic Carbon Nitride via XRD and Neutron Diffraction
What Makes a Strong Organic Electron Donor (or Acceptor)?
Abstract
Organic electron donors are of importance for a number of applications. However, the factors that are essential for a directed design of compounds with desired reduction power are not clear. Here, we analyze these factors in detail. The intrinsic reduction power, which neglects the environment, has to be separated from extrinsic (e.g., solvent) effects. This power could be quantified by the gas-phase ionization energy. The experimentally obtained redox potentials in solution and the calculated ionization energies in a solvent (modeled with the conductor-like screening model (COSMO)) include both intrinsic and extrinsic factors. An increase in the conjugated π-system of organic electron donors leads to an increase in the intrinsic reduction power, but also decreases the solvent stabilization. Hence, intrinsic and extrinsic effects compete against each other; generally the extrinsic effects dominate. We suggest a simple relationship between the redox potential in solution and the gas-phase ionization energy and the volume of an organic electron donor. We finally arrive at formulas that allow for an estimate of the (gas-phase) ionization energy of an electron donor or the (gas-phase) electron affinity of an electron acceptor from the measured redox potentials in solution. The formulas could be used for neutral organic molecules with no or only small static dipole moment and relatively uniform charge distribution after oxidation/reduction.

Size matters: A relationship between the gas-phase ionization energy and the experienced redox potential in solution is derived. The results highlight the importance of molecular volume in determining the reduction potential in solution (see figure).
How to Improve Spermbot Performance
Spermbots are biocompatible hybrid machines that consist of microtubes which are propelled by single spermatozoa and have promising features for powering nano and microdevices. This article presents three approaches on how to improve the performance of such spermbots. First, 20 μm microtubes produce faster spermbots compared to the previously reported 50 μm long microtubes. Furthermore, biofunctionalization by microcontact printing and surface chemistry of biomolecules on the inner tube surface improve the coupling efficiency between sperm cell and microtube, and the addition of caffeine results in a speed boost of the sperm-driven micromotor.

Improved spermbot performance is demonstrated by biofunctionalization of the inner tube surface, shorter tube design, and caffeine addition. Firstly, spermbot velocity is improved by the use of shorter microtubes; secondly, better coupling efficiency is achieved by binding of fibronectin inside the microtube; and finally, caffeine addition gives a temporary speed boost to the spermbot.
Hydrogen Evolution: Hybrid Z-Scheme Using Photosystem I and BiVO4 for Hydrogen Production (Adv. Funct. Mater. 16/2015)

In the Z-scheme of photosynthesis, two photosynthetic proteins, photosystem II and photosystem I, excite electrons step-wise using the light energy. On page 2369, K. T. Nam and team report a hybrid Z-scheme using photosystem I and a BiVO4 semiconductor. In this study, step-wise charge separation in photosystem I and BiVO4 enables the production of hydrogen from only water under visible light, for the first time.
Back Cover: The Mechanism of CO and CO2 Hydrogenation to Methanol over Cu-Based Catalysts (ChemCatChem 7/2015)

The cover picture shows four different pathways for methanol synthesis over Cu-based catalysts depending on the feed composition and the presence of the Zn-promoter. In their Full Paper, F. Studt et al. present a DFT-based microkinetic model and consistent experimental data that explain the Janus-faced character of Cu in COx hydrogenation allowing only one path, CO or CO2 hydrogenation, to be effective at a time. The industrial Zn-promoter accelerates CO2 conversion over Cu/ZnO, but can simultaneously act as a poison for CO hydrogenation. On the other hand, CO2 in the feed was found to poison unpromoted catalysts such as Cu/MgO, which was highly efficient for CO hydrogenation. The article highlighted by this cover can be found on p. 1105 ff. of Issue 7, 2015.
A Consistent Reaction Scheme for the Selective Catalytic Reduction of Nitrogen Oxides with Ammonia

Effects of Surface Defects on Photocatalytic H2O2 Production by Mesoporous Graphitic Carbon Nitride under Visible Light Irradiation

Mechanistic Insights into the Reduction of CO2 on Tin Electrodes using in Situ ATR-IR Spectroscopy

Catalytic Hydroboration of Carbonyl Derivatives, Imines, and Carbon Dioxide

Noble metal-free hydrogen evolution catalysts for water splitting
DOI: 10.1039/C4CS00448E, Review Article
This review summarizes the recent research efforts toward noble metal-free hydrogen evolution electrocatalysts.
The content of this RSS Feed (c) The Royal Society of Chemistry
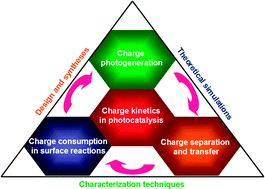
Steering charge kinetics in photocatalysis: intersection of materials syntheses, characterization techniques and theoretical simulations
DOI: 10.1039/C5CS00064E, Review Article
This review outlines the recent progress on probing and steering charge kinetics toward designing highly efficient photocatalysts.
The content of this RSS Feed (c) The Royal Society of Chemistry
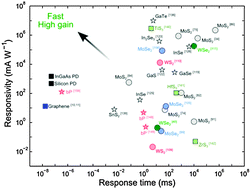
Photocurrent generation with two-dimensional van der Waals semiconductors
DOI: 10.1039/C5CS00106D, Review Article
We review photodetectors based on transition metal dichalcogenides, novel van der Waals materials, black phosphorus, and heterostructures.
The content of this RSS Feed (c) The Royal Society of Chemistry
Editorial: Sustainable Chemistry for Energizing the Planet
Combined biomass valorization and hydrogen production in a photoelectrochemical cell

Nature Chemistry 7, 328 (2015). doi:10.1038/nchem.2194
Authors: Hyun Gil Cha & Kyoung-Shin Choi
Photoelectrochemical water-splitting produces hydrogen at the cathode and oxygen at the anode. The anode reaction is, however, kinetically unfavourable. Now, reduction of water at the cathode has been combined with oxidation of 5-hydroxymethylfurfural at the anode resulting in a photoelectrochemical cell that produces fuel and a useful platform chemical.
Visible light-driven efficient overall water splitting using p-type metal-nitride nanowire arrays
Article
Solar water splitting for hydrogen generation may be a future source of renewable energy. Here, the authors demonstrate that controlled p -type doping of metal-nitride nanowires can eradicate surface potential barriers and promotes stable stoichiometric dissociation of water under visible light.
Nature Communications doi: 10.1038/ncomms7797
Authors: M. G. Kibria, F. A. Chowdhury, S. Zhao, B. AlOtaibi, M. L. Trudeau, H. Guo, Z. Mi
Energy transfer pathways in semiconducting carbon nanotubes revealed using two-dimensional white-light spectroscopy
Article
Thin films of carbon nanotubes are been considered for energy harvesting and optoelectronic devices but their energy transfer pathways are largely unknown. Here, Mehlenbacher et al . use two-dimensional white-light spectroscopy to investigate the ultrafast energy redistribution in carbon nanotube films.
Nature Communications doi: 10.1038/ncomms7732
Authors: Randy D. Mehlenbacher, Thomas J. McDonough, Maksim Grechko, Meng-Yin Wu, Michael S. Arnold, Martin T. Zanni
Efficient light emission from inorganic and organic semiconductor hybrid structures by energy-level tuning
Article
Hybrid inorganic-organic structures can overcome the limits of inorganic semiconductor light emitting devices but the energy level offset is an obstacle. Here, Schlesinger et al. lower the ZnO work function with an organometallic donor monolayer and enhance the radiative emission of the hybrid structure.
Nature Communications doi: 10.1038/ncomms7754
Authors: R. Schlesinger, F. Bianchi, S. Blumstengel, C. Christodoulou, R. Ovsyannikov, B. Kobin, K. Moudgil, S. Barlow, S. Hecht, S.R. Marder, F. Henneberger, N. Koch
Stimuli-Responsive Metal–Ligand Assemblies
Back Cover: Highly Enantioselective Transfer Hydrogenation of Ketones with Chiral (NH)2P2 Macrocyclic Iron(II) Complexes (Angew. Chem. Int. Ed. 17/2015)

Setting sail towards new horizons is the iron(II)-catalyzed asymmetric transfer hydrogenation of C![[DOUBLE BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8fe.gif) O bonds. With the robust, highly active N2P2 macrocyclic catalysts described by A. Mezzetti and co-workers in their Communication on page 5171 ff., a wide scope of substrates can be hydrogenated, obtaining high yields and enantioselectivities by literally paying the iron price.
O bonds. With the robust, highly active N2P2 macrocyclic catalysts described by A. Mezzetti and co-workers in their Communication on page 5171 ff., a wide scope of substrates can be hydrogenated, obtaining high yields and enantioselectivities by literally paying the iron price.
High Catalytic Activity of Heteropolynuclear Cyanide Complexes Containing Cobalt and Platinum Ions: Visible-Light Driven Water Oxidation
Abstract
A near-stoichiometric amount of O2 was evolved as observed in the visible-light irradiation of an aqueous buffer (pH 8) containing [RuII(2,2′-bipyridine)3] as a photosensitizer, Na2S2O8 as a sacrificial electron acceptor, and a heteropolynuclear cyanide complex as a water-oxidation catalyst. The heteropolynuclear cyanide complexes exhibited higher catalytic activity than a polynuclear cyanide complex containing only CoIII or PtIV ions as C-bound metal ions. The origin of the synergistic effect between Co and Pt ions is discussed in relation to electronic and local atomic structures of the complexes.

A platinum assist: A near-stoichiometric amount of O2 was evolved as observed in the visible-light irradiation of an aqueous buffer (pH 8) containing [RuII(2,2′-bipyridine)3] as a photosensitizer, Na2S2O8 as a sacrificial electron acceptor, and a heteropolynuclear cyanide complex as a water-oxidation catalyst. The synergistic effect between the Co and Pt ions was confirmed to facilitate the water-oxidation catalysis by the heteropolynuclear complex.
Ammonia Activation by a Nickel NCN-Pincer Complex featuring a Non-Innocent N-Heterocyclic Carbene: Ammine and Amido Complexes in Equilibrium
Abstract
A Ni0-NCN pincer complex featuring a six-membered N-heterocyclic carbene (NHC) central platform and amidine pendant arms was synthesized by deprotonation of its NiII precursor. It retained chloride in the square-planar coordination sphere of nickel and was expected to be highly susceptible to oxidative addition reactions. The Ni0 complex rapidly activated ammonia at room temperature, in a ligand-assisted process where the carbene carbon atom played the unprecedented role of proton acceptor. For the first time, the coordinated (ammine) and activated (amido) species were observed together in solution, in a solvent-dependent equilibrium. A structural analysis of the Ni complexes provided insight into the highly unusual, non-innocent behavior of the NHC ligand.

Fast and reversible ligand-assisted ammonia activation on a nickel-NCN pincer complex featuring a non-innocent N-heterocyclic carbene produced a solvent-dependent equilibrium mixture of ammine and amide complexes (see scheme, Dipp=2,6-diisopropylphenyl). These species were present in solution in comparable proportions, and EXSY NMR experiments as well as exchange with ND3 further supported the equilibrium.
High Catalytic Activity of Heteropolynuclear Cyanide Complexes Containing Cobalt and Platinum Ions: Visible-Light Driven Water Oxidation

Nearly stoichiometric amounts of O2 are evolved on visible-light irradiation of an aqueous buffer (pH 8) containing [RuII(2,2′-bipyridine)3], Na2S2O8, and a heteropolynuclear cyanide complex containing Co and Pt ions as a photosensitizer, a sacrificial electron acceptor, and a water oxidation catalyst, respectively, as reported by Y. Yamada, S. Fukuzumi, et al. in their Communication (DOI: 10.1002/anie.201501116). A synergistic effect between the Co and Pt ions was confirmed for the water oxidation catalysis by the heteropolynuclear complex.
Micromotor-Based Energy Generation
Abstract
A micromotor-based strategy for energy generation, utilizing the conversion of liquid-phase hydrogen to usable hydrogen gas (H2), is described. The new motion-based H2-generation concept relies on the movement of Pt-black/Ti Janus microparticle motors in a solution of sodium borohydride (NaBH4) fuel. This is the first report of using NaBH4 for powering micromotors. The autonomous motion of these catalytic micromotors, as well as their bubble generation, leads to enhanced mixing and transport of NaBH4 towards the Pt-black catalytic surface (compared to static microparticles or films), and hence to a substantially faster rate of H2 production. The practical utility of these micromotors is illustrated by powering a hydrogen–oxygen fuel cell car by an on-board motion-based hydrogen and oxygen generation. The new micromotor approach paves the way for the development of efficient on-site energy generation for powering external devices or meeting growing demands on the energy grid.

Fuel metal jacket: Micromotors give enhanced energy generation by the movement of Pt-black/Ti Janus microparticles in liquid-phase chemical fuel. The autonomous motion of these micromotors leads to enhanced mixing and transport of NaBH4 fuel compared to static microparticles or films, and hence to a substantially faster hydrogen-generation rate. The practical utility is illustrated by powering a hydrogen–oxygen fuel cell car.
Criticality of metals and metalloids [Sustainability Science]
[Feature] Deepwater Horizon: After the oil
[Editors' Choice] How smoking makes infection worse
Activation of Water in Titanium Dioxide Photocatalysis by Formation of Surface Hydrogen Bonds: An In Situ IR Spectroscopy Study
Abstract
The hole-driving oxidation of titanium-coordinated water molecules on the surface of TiO2 is both thermodynamically and kinetically unfavorable. By avoiding the direct coordinative adsorption of water molecules to the surface Ti sites, the water can be activated to realize its oxidation. When TiO2 surface is covered by the H-bonding acceptor F, the first-layer water adsorption mode is switched from Ti coordination to a dual H-bonding adsorption on adjacent surface F sites. Detailed in situ IR spectroscopy and isotope-labeling studies reveal that the adsorbed water molecules by dual H-bonding can be oxidized to O2 even in the absence of any electron scavengers. Combined with theoretical calculations, it is proposed that the formation of the dual H-bonding structure can not only enable the hole transfer to the water molecules thermodynamically, but also facilitate kinetically the cleavage of O![[BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) H bonds by proton-coupled electron transfer process during water oxidation.
H bonds by proton-coupled electron transfer process during water oxidation.

Switching the water adsorption mode on TiO2 to dual H-bonding by the presence of fluorine atoms at the surface not only thermodynamically enables the hole transfer to the water molecules, but also facilitates the proton-coupled electron transfer during water oxidation. This phenomenon is established by IR spectroscopy studies and calculations.
A Metal-Free N-Annulated Thienocyclopentaperylene Dye: Power Conversion Efficiency of 12 % for Dye-Sensitized Solar Cells
Abstract
Reported are two highly efficient metal-free perylene dyes featuring N-annulated thienobenzoperylene (NTBP) and N-annulated thienocyclopentaperylene (NTCP), which are coplanar polycyclic aromatic hydrocarbons. Without the use of any coadsorbate, the metal-free organic dye derived from the NTCP segment was used for a dye-sensitized solar cell which attained a power conversion efficiency of 12 % under an irradiance of 100 mW cm−2, simulated air mass global (AM1.5G) sunlight.

Power trip: A perylene dye derived from N-annulated thienocyclopentaperylene, which is characterized by a low-energy gap and a high electron injection yield, was synthesized for dye-sensitized solar cells. A high power conversion efficiency of 12 %, at an irradiance of the AM1.5G sunlight, was achieved. This efficiency is the highest achieved thus far by using just a metal-free organic dye.
An allosteric photoredox catalyst inspired by photosynthetic machinery
Article
Photosynthetic systems regulate light harvesting via structural and electronic control of antenna proteins. Here, the authors report a light-harvesting antenna/reaction centre mimic that can be allosterically regulated using mild and redox-inactive inputs, via a coordination framework with hemilabile ligands.
Nature Communications doi: 10.1038/ncomms7541
Authors: Alejo M. Lifschitz, Ryan M. Young, Jose Mendez-Arroyo, Charlotte L. Stern, C. Michael McGuirk, Michael R. Wasielewski, Chad A. Mirkin
Electride support boosts nitrogen dissociation over ruthenium catalyst and shifts the bottleneck in ammonia synthesis
Article
Development of catalysts that enhance dissociation of the nitrogen–nitrogen triple bond will reduce costs of ammonia production. Here, the authors study ammonia synthesis over a ruthenium loaded electride catalyst and show that the rate-determining step is shifted to nitrogen–hydrogen bond formation.
Nature Communications doi: 10.1038/ncomms7731
Authors: Masaaki Kitano, Shinji Kanbara, Yasunori Inoue, Navaratnarajah Kuganathan, Peter V. Sushko, Toshiharu Yokoyama, Michikazu Hara, Hideo Hosono