06 Mar 12:02
Chem. Commun., 2018, 54,3122-3125
DOI: 10.1039/C8CC00209F, Communication
Chengyong Li, Yu Zhao, Lei He, Rijian Mo, Hongli Gao, Chunxia Zhou, Pengzhi Hong, Shengli Sun, Guigen Zhang
A mussel-inspired new interfacial ionic rectification device is fabricated using porous anodic alumina nanochannels and graphene oxide via dopamine polymerization.
The content of this RSS Feed (c) The Royal Society of Chemistry

19 Feb 12:48
by Erica Y. Tsai, Richard Y. Liu, Yang Yang and Stephen L. Buchwald

Journal of the American Chemical Society
DOI: 10.1021/jacs.7b12271

24 Oct 08:12
by Zhichao Lu, Junbin Han, Otome E. Okoromoba, Naoto Shimizu, Hideki Amii, Cláudio F. Tormena, Gerald B. Hammond and Bo Xu

Organic Letters
DOI: 10.1021/acs.orglett.7b02829

04 Jan 17:08
by Wei Chen, Ya-Li Bai, Yong-Chun Luo and Peng-Fei Xu

Organic Letters
DOI: 10.1021/acs.orglett.6b03542

28 Nov 13:59
Chem. Commun., 2016, Accepted Manuscript
DOI: 10.1039/C6CC07874E, Communication
Nitin T. Patil, Indradweep Chakrabarty, Suleman M Inamdar, Manjur Oyasim Akram, Amol B. Gade, Subhrashis Banerjee, Saibal Bera
A Pt-catalyzed, highly regioselective, reaction between N-allenamides and imino-alkynes leading to pyrrolo[1,2-a]indoles is described. This represents the first example of [3+2]-annulation of Pt-bound azomethine ylides with the distal C=C bond...
The content of this RSS Feed (c) The Royal Society of Chemistry

大河弯了 and -1 others like this
24 Nov 09:34
Chem. Commun., 2016, 52,14462-14465
DOI: 10.1039/C6CC08385D, Communication
Pradip N. Bagle, Manoj V. Mane, K. Vanka, Dinesh R. Shinde, Samir R. Shaikh, Rajesh G. Gonnade, Nitin T. Patil
A co-operative Au/Ag catalyst system has been developed to utilize N-allenamides as nucleophilic enal equivalents for the interceptive capturing of incipient carbocations generated through [small pi]-acid-triggered imino-alkyne cyclization.
The content of this RSS Feed (c) The Royal Society of Chemistry

醉卧红尘 and -1 others like this
24 Nov 09:16
by Silvia González-Pelayo, Luis A. López
Abstract
An efficient, metal-free, silicon–hydrogen bond functionalization based on the microwave-assisted reaction of readily available enynones and silanes is reported. This process seemingly proceeds through a 2-furyl carbene species, a particularly elusive intermediate. Preliminary studies on the metal-free oxygen–hydrogen and nitrogen–hydrogen bond functionalization of representative alcohols, azoles and sulfonamides are also provided.

14 Nov 09:49
by José M. González, Kilian Muñiz

José Barluenga, emeritus professor at the University of Oviedo, passed away on September 7, 2016. Barluenga was one of the leading Spanish chemists of his generation, with research interests including metal-mediated activation of alkenes and alkynes, β-functionalized organolithium compounds, iodination reactions, asymmetric Diels–Alder reactions, carbene complexes, and cross-coupling reactions.
14 Jul 09:07
by Wei-Feng Zheng, Pranjal Protim Bora, Gui-Jun Sun and Qiang Kang

Organic Letters
DOI: 10.1021/acs.orglett.6b01731

13 May 17:47
by Sandeep Suryabhan Gholap, Masanori Takimoto, Zhaomin Hou
Abstract
The alkylative carboxylation of allenamide catalyzed by an N-heterocyclic carbene (NHC)–copper(I) complex [(IPr)CuCl] with CO2 and dialkylzinc reagents was investigated. The reaction of allenamides with dialkylzinc reagents (1.5 equiv) and CO2 (1 atm.) proceeded smoothly in the presence of a catalytic quantity of [(IPr)CuCl] to afford (Z)-α,β-dehydro-β-amino acid esters in good yields. The reaction is regioselective, with the alkyl group introduced onto the less hindered γ-carbon, and the carboxyl group introduced onto the β-carbon atom of the allenamides. The first step of the reaction was alkylative zincation of the allenamides to give an alkenylzinc intermediate followed by nucleophilic addition to CO2. A variety of cyclic and acyclic allenamides were found to be applicable to this transformation. Dialkylzinc reagents bearing β-hydrogen atoms, such as Et2Zn or Bu2Zn, also gave the corresponding alkylative carboxylation products without β-hydride elimination. The present methodology provides an easy route to alkyl-substituted α,β-dehydro-β-amino acid ester derivatives under mild reaction conditions with high regio- and stereoselectivtiy.

Alkylation and CO2 Incorporation: The alkylative carboxylation of allenamides with dialkylzinc reagents and carbon dioxide in the presence of a catalytic amount of an N-Heterocyclic Carbene (NHC)–copper complex was achieved. The reactions proceed through Cu-catalyzed carbozincation of allenamides with dialkylzinc reagents and the subsequent nucleophilic addition of the resulting alkenyl zinc species to CO2 (see scheme).
05 May 09:09
Chem. Soc. Rev., 2016, 45,4533-4551
DOI: 10.1039/C6CS00171H, Review Article
R. J. Harris, R. A. Widenhoefer
This review highlights recent advances made in the synthesis of cationic gold carbene complexes and in the direct detection of cationic intermediates generated via gold-catalysed enyne cycloaddition.
The content of this RSS Feed (c) The Royal Society of Chemistry

18 Mar 12:40
by Jairo González, Javier Santamaría, Ángel L. Suárez-Sobrino, Alfredo Ballesteros
Abstract
A three-component gold(I)-catalyzed synthesis of 2-imidazoyl-1-pyrazolylbenzenes from 1-propargyl-1H-benzotriazoles is described here. Initially the benzotriazole derivative suffers an intramolecular 5-endo-dig cyclization to form a triazapentalene. This dipolar compound is able to perform an intermolecular and regioselective attack to a gold-activated alkyne. After triazole breakage, the pyrazole ring and an α-imino gold carbene complex are formed. Finally, the iminocarbene is captured by a nitrile to form an imidazole ring.

17 Mar 18:43
by Manuel Plaza, M. Carmen Pérez-Aguilar, Carlos Valdés
Abstract
The reactions between alkenylboronic acids and tosylhydrazones derived from substituted cyclohexanones lead to the construction of disubstituted cyclohexanes with total regio- and stereoselectivity. In these transition-metal-free processes, a Csp3−Csp2 and Csp3−H bond are formed on the same carbon atom. The stereoselective reaction is general for 2-, 3-, and 4-substituted cyclohexanone tosylhydrazones, as well as for 2-substituted cyclopentanones. However, no stereoselectivity is observed for acyclic derivatives. DFT computational modeling suggests that the stereoselectivity of the reaction is determined by the approach of the boronic acid to the diazocyclohexane on its most stable chair conformation through an equatorial trajectory.

An orphan transformation: Two bonds on the same carbon atom Csp3−Csp2 and Csp3−H bonds are formed in a diastereoselective manner by reaction of tosylhydrazones of substituted cyclic ketones with alkenyl boronic acids. This transition-metal-free reductive coupling represents a solution for a previously unavailable stereoselective transformation of carbonyl compounds (see scheme).
23 Feb 09:42
Chem. Soc. Rev., 2016, 45,4567-4589
DOI: 10.1039/C5CS00929D, Review Article
Weiwei Zi, F. Dean Toste
This review highlights advances in enantioselective gold catalysis over the past five years.
The content of this RSS Feed (c) The Royal Society of Chemistry

22 Feb 11:55
by Xianjie Fang
Nitriles and alkenes are important synthetic intermediates with complementary reactivity that play a central role in the preparation of materials, pharmaceuticals, cosmetics, and agrochemicals. Here, we report a nickel-catalyzed transfer hydrocyanation reaction between a wide range (60 examples) of alkyl nitriles and alkenes. This strategy not only overcomes the toxicity challenge posed by the use of HCN in traditional approaches, but also encompasses distinct chemical advances, including retro-hydrocyanation and anti-Markovnikov regioselectivity. In a broader context, this work highlights an approach to the reversible hydrofunctionalization of alkenes through thermodynamically controlled transfer reactions to circumvent the use of volatile and hazardous reagents in the laboratory.
Authors: Xianjie Fang, Peng Yu, Bill Morandi
12 Feb 09:52
by Paulo H. S. Paioti, Khalil A. Abboud and Aaron Aponick

Journal of the American Chemical Society
DOI: 10.1021/jacs.5b13387

12 Feb 09:50
by Quirin M. Kainz
Despite a well-developed and growing body of work in copper catalysis, the potential of copper to serve as a photocatalyst remains underexplored. Here we describe a photoinduced copper-catalyzed method for coupling readily available racemic tertiary alkyl chloride electrophiles with amines to generate fully substituted stereocenters with high enantioselectivity. The reaction proceeds at –40°C under excitation by a blue light-emitting diode and benefits from the use of a single, Earth-abundant transition metal acting as both the photocatalyst and the source of asymmetric induction. An enantioconvergent mechanism transforms the racemic starting material into a single product enantiomer.
Authors: Quirin M. Kainz, Carson D. Matier, Agnieszka Bartoszewicz, Susan L. Zultanski, Jonas C. Peters, Gregory C. Fu
15 Jan 11:21
by Fang-Lin Zhang
Proximity-driven metalation has been extensively exploited to achieve reactivity and selectivity in carbon–hydrogen (C–H) bond activation. Despite the substantial improvement in developing more efficient and practical directing groups, their stoichiometric installation and removal limit efficiency and, often, applicability as well. Here we report the development of an amino acid reagent that reversibly reacts with aldehydes and ketones in situ via imine formation to serve as a transient directing group for activation of inert C–H bonds. Arylation of a wide range of aldehydes and ketones at the β or γ positions proceeds in the presence of a palladium catalyst and a catalytic amount of amino acid. The feasibility of achieving enantioselective C–H activation reactions using a chiral amino acid as the transient directing group is also demonstrated.
Authors: Fang-Lin Zhang, Kai Hong, Tuan-Jie Li, Hojoon Park, Jin-Quan Yu
12 Jan 11:33
Chem. Commun., 2016, 52,2905-2908
DOI: 10.1039/C5CC09529H, Communication
Alberto Ballesteros, Pablo Moran-Poladura, Jose M. Gonzalez
A synergistic approach to the catalytic intermolecular [small alpha]-addition of aldehydes across allenamides using gold(I) and proline derivatives.
The content of this RSS Feed (c) The Royal Society of Chemistry

11 Dec 11:37
by Joshua H. Baraban
Transition state theory is central to our understanding of chemical reaction dynamics. We demonstrate a method for extracting transition state energies and properties from a characteristic pattern found in frequency-domain spectra of isomerizing systems. This pattern—a dip in the spacings of certain barrier-proximal vibrational levels—can be understood using the concept of effective frequency, ωeff. The method is applied to the cis-trans conformational change in the S1 state of C2H2 and the bond-breaking HCN-HNC isomerization. In both cases, the barrier heights derived from spectroscopic data agree extremely well with previous ab initio calculations. We also show that it is possible to distinguish between vibrational modes that are actively involved in the isomerization process and those that are passive bystanders.
Authors: Joshua H. Baraban, P. Bryan Changala, Georg Ch. Mellau, John F. Stanton, Anthony J. Merer, Robert W. Field
04 Dec 16:05
by Cristina Hernández-Díaz, Eduardo Rubio, José M. González
Abstract
A protocol involving a gold(I)-catalyzed reaction to elaborate cyclic frames by selective trimerization reactions of N-allenylsulfonamides is reported. Efficiency in the formation of the target [2+2+2] products depends on the concentration of the reagents, the nature of the ancillary ligand on the gold catalyst, and, to a significant extent, on the selection of a low reaction temperature. This singular catalytic allene cycloaddition gives adducts in high isolated yields. In addition to representing a new gold-catalyzed reaction, the reported allenamide cyclotrimerization offers a significant extension to the scant number of allenes that have effectively entered this cyclization mode by use of other metal-catalyzed reactions. Upon aromatization of the assembled adducts, the overall reaction matches the outcome of related and widely investigated alkyne trimerization reactions.

Granting gilding access to triskelium-like molecules: The first examples of a gold-catalyzed [2+2+2]-cycloaddition reaction of tosylallenamides are presented. Upon ligand screening, conditions have been devised to favor the direct assembly of molecules with this particular shape.
16 Nov 16:02
by Maximilian Joost, Abderrahmane Amgoune, Didier Bourissou
Abstract
For a while, the reactivity of gold complexes was largely dominated by their Lewis acid behavior. In contrast to the other transition metals, the elementary steps of organometallic chemistry—oxidative addition, reductive elimination, transmetallation, migratory insertion—have scarcely been studied in the case of gold or even remained unprecedented until recently. However, within the last few years, the ability of gold complexes to undergo these fundamental reactions has been unambiguously demonstrated, and the reactivity of gold complexes was shown to extend well beyond π-activation. In this Review, the main achievements described in this area are presented in a historical context. Particular emphasis is set on mechanistic studies and structure determination of key intermediates. The electronic and structural parameters delineating the reactivity of gold complexes are discussed, as well as the remaining challenges.

Sitting on a gold mine? In the past few years, the reactivity of gold complexes has been extended well beyond Lewis acid behavior and electrophilic activation of π-substrates. Elementary steps considered highly unlikely, if not impossible, for gold complexes have been unambiguously demonstrated, offering new perspectives in gold catalysis.
23 Oct 15:44
by Yidong Wang, Peichao Zhang, Deyun Qian, Junliang Zhang

A gold(I)-catalyzed intermolecular annulation of N-allenamides at the proximal C![[DOUBLE BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8fe.gif) C bond has been achieved. In their Communication on page 14849 ff., J. Zhang et al. show that both enantiomers of the products are obtained with high regio-, diastereo-, and enantioselectivity by using either diastereomer of a chiral phosphoramidite as ligand. In the picture, the enantiomers of the products are easily controlled by the magician’s hands with the aid of the gold rings on the fingers.
C bond has been achieved. In their Communication on page 14849 ff., J. Zhang et al. show that both enantiomers of the products are obtained with high regio-, diastereo-, and enantioselectivity by using either diastereomer of a chiral phosphoramidite as ligand. In the picture, the enantiomers of the products are easily controlled by the magician’s hands with the aid of the gold rings on the fingers.
24 Sep 14:00
by Jairo González, Javier Santamaría, Alfredo Ballesteros
Abstract
A gold(I)-catalyzed synthesis of indanones from trimethylsilylacetylenes and acylsilanes is presented. The reaction is initiated through a synergistic acylsilane activation–gold acetylide formation and involves consecutive alkyne σ-gold(I) addition, π-activation, and 1,2-migration of a silyl group. Studies performed on the reaction mechanism allowed to establish the nature of the silyl migrating group and invoke the participation of a gold(I) carbenoid intermediate. The reaction is completed by a gold(I) C![[BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) H functionalization step.
H functionalization step.

The elegant way: A gold(I)-catalyzed synthesis of indanones from trimethylsilylacetylenes and acylsilanes was developed. The reaction involves a synergistic acylsilane activation–gold acetylide formation and consecutive alkyne σ-gold(I) addition, π-activation, and 1,2-silyl migration. Mechanistic studies suggest the participation of a gold(I) carbenoid intermediate.
02 Sep 10:30
by María J. González, Javier González, Luís A. López, Rubén Vicente
Abstract
The zinc-catalyzed reaction of cyclopropenes with alkenes leading to vinylcyclopropane derivatives is reported. A broad range of alkenes (including highly substituted or functionalized alkenes) is compatible with this protocol. On the basis of trapping experiments and computational studies, this cyclopropanation reaction is proposed to proceed through initial formation of an electrophilic zinc vinyl carbenoid intermediate, which may be involved in a concerted cyclopropanation reaction. The reported protocol represents an unprecedented and simple strategy for the catalytic generation of zinc vinyl carbenoids, which are promising intermediates in organic synthesis.

Open and close! The title reaction provides a convenient and general route to relevant vinylcyclopropane derivatives. Mechanistic studies support the participation of a zinc vinylcarbene intermediate, which may be subsequently involved in a concerted cyclopropanation reaction. This method represents a step towards identifying suitable precursors for the catalytic generation of zinc carbenoids.
04 Mar 10:44
by Raquel de la Campa, Irene Ortín, Darren J. Dixon
Abstract
A catalytic asymmetric aldol addition/cyclization reaction of unactivated ketones with isocyanoacetate pronucleophiles has been developed. A quinine-derived aminophosphine precatalyst and silver oxide were found to be an effective binary catalyst system and promoted the reaction to afford chiral oxazolines possessing a fully substituted stereocenter with good diastereoselectivities and excellent enantioselectivities.

A stereoselective aldol reaction of unactivated ketones and isocyanoacetate pronucleophiles is catalyzed by a binary catalyst system consisting of an aminophosphine precatalyst and silver(I) oxide and affords oxazoline products with a fully substituted β-carbon atom. The reaction is efficient and broad in scope and proceeds with high diastereo- and enantioselectivity.
02 Mar 16:46
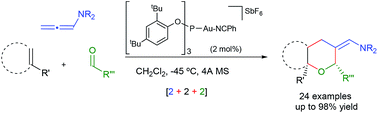
Chem. Sci., 2015, 6,2903-2908
DOI: 10.1039/C5SC00295H, Edge Article

Open Access
Helio Faustino, Ivan Varela, Jose L. Mascarenas, Fernando Lopez
A novel fully intermolecular gold-catalyzed [2 + 2 + 2] cycloaddition involving an allenamide, an alkene and an aldehyde provides a straightforward entry to tetrahydropyrans.
The content of this RSS Feed (c) The Royal Society of Chemistry

26 Feb 18:40
by Vinayak Vishnu Pagar, Rai-Shung Liu
Abstract
Gold-catalyzed cycloadditions of ethyl diazoacetate, nitrosoarenes, and vinyldiazo carbonyl species to yield isoxazolidine derivatives stereoselectively are described. Treatment of these isoxazolidine products with the same catalyst results in a novel 1,2-H shift/[3,3] rearrangement to give benzo[b]azepine compounds. The mechanism of this skeletal rearrangement is elucidated with deuterium-labeling experiments.

A little shifty: The title reaction yields isoxazolidine derivatives stereoselectively. Treatment of these isoxazolidine products with the same catalyst results in a novel 1,2-H shift/[3,3] rearrangement to give benzo[b]azepine compounds. The mechanism of this skeletal rearrangement is elucidated with deuterium-labeling experiments. FG=functional group.
19 Feb 18:57
by Minqiang Jia and Marco Bandini

ACS Catalysis
DOI: 10.1021/cs501902v

21 Jan 11:45
by Pablo Morán-Poladura, Eduardo Rubio, José M. González
Abstract
The cycloisomerization reaction of 1-(iodoethynyl)-2-(1-methoxyalkyl)arenes and related 2-alkyl-substituted derivatives gives the corresponding 3-iodo-1-substituted-1H-indene under the catalytic influence of IPrAuNTf2 [IPr=1,3-bis(2,6-diisopropyl)phenylimidazol-2-ylidene; NTf2=bis(trifluoromethanesulfonyl)imidate]. The reaction takes place in 1,2-dichloroethane at 80 °C, and the addition of ttbp (2,4,6-tri-tert-butylpyrimidine) is beneficial to accomplish this new transformation in high yield. The overall reaction implies initial assembly of an intermediate gold vinylidene upon alkyne activation by gold(I) and a 1,2-iodine-shift. Deuterium labeling and crossover experiments, the magnitude of the recorded kinetic primary isotopic effect, and the results obtained from the reaction of selected stereochemical probes strongly provide support for concerted insertion of the benzylic C![[BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) H bond into gold vinylidene as the step responsible for the formation of the new carbon–carbon bond.
H bond into gold vinylidene as the step responsible for the formation of the new carbon–carbon bond.

Golden dance: The cycloisomerization reaction of 1-(iodoethynyl)-2-(1-methoxyalkyl)arenes gives the corresponding 3-iodo-1-substituted-1H-indene. Gold(I) catalysis triggers a selective intramolecular carbon–carbon bond-forming event, which involves the insertion of benzylic C![[BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) H bonds in the catalytically assembled gold(I) iodovinylidene intermediate.
H bonds in the catalytically assembled gold(I) iodovinylidene intermediate.


















![[DOUBLE BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8fe.gif) C bond has been achieved. In their Communication on
C bond has been achieved. In their Communication on ![[BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) H functionalization step.
H functionalization step.


 Open Access
Open Access


