A. Kachmar
Shared posts
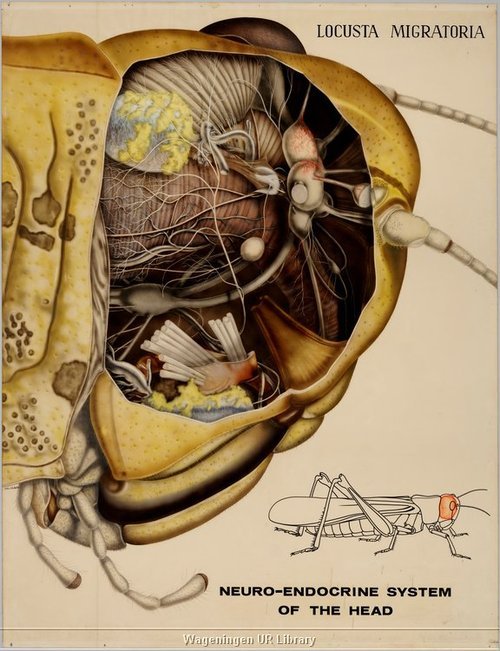
Photo
A. KachmarI'm moving to https://www.inoreader.com/u/kachmar
I won't be active on here anymore.

15+ Hilarious Examples Of Cat Logic
Don’t Sit On The Keyboard?

Seriously? Cat Logic

Finally Found A Perfect Place To Sit

Cat Logic

Got My Cat A Bed For Christmas

Cat Logic

You’re Doing It Wrong

This Is My Friends Cat Eating Dinner

Double Bed

Cat Logic

Cat Logic

Still Trying To Figure Out The Logic Behind This One

Cat Logic

Cat Logic Always Works

This Is How My Cat Eats

malformalady: Sculpture by artist Emil Melmoth. Of his work...


Sculpture by artist Emil Melmoth. Of his work Melmoth says “Death is living forever on my mind, a self-portrait of so many changes i had recently and how i see myself inside. “Death as the Arcane XIII or Transfiguration”(work in progress)
owlturdcomix: That’ll look cute. image | twitter | facebook
'I don't want to be interesting. I want to be good.'
A. KachmarThe music is a bit distracting
asylum-art-2: Fire Paintings Draws With Flames And Soot by...










Fire Paintings Draws With Flames And Soot by Steven Spazuk
More info: spazuk.com | Youtube | Facebook
Steven Spazuk is a Canada-based artist who uses candle soot to create elegant drawings. After depositing soot on his media with a candle or torch, he etches lines and patterns in the soot with pencils and feathers.
Though Spazuk has spent the last 14 years developing and perfecting his soot painting technique, the creation process always has an element of random spontaneity and improvisation. via: boredpanda
Highlights from Artist Tatsuya Tanaka’s Daily Miniature Photo Project

Photographer and art director Tatsuya Tanaka has a fascination with all things tiny and has an uncanny ability to repurpose everyday objects as set pieces or tools for the inhabitants of his miniature world. For his project Miniature Calendar, Tanaka has been stretching his imagination to its limits nearly every day for the last four years. A tape dispenser becomes the bar for a restaurant, a circuit board is suddenly a rice paddy field, and the notes of a musical score become the hurdles for a track race. Individually, the photos might invoke a smile or chuckle as you get the joke, but when viewed collectively they morph into a fascinating study on Tanaka’s breadth of creativity.
New photos from Miniature Calendar are published every day on Instagram and Facebook. Tanaka also published a book of earlier miniature photos in a book titled Miniature Life. (via Spoon & Tamago)






ultrafitx: Always have room for melon in your diet. 😏
Hold This Magnet In Your Hand and It Stops Being a Magnet
"وطن المرء ليس مكان ولادته و لكنه المكان الذي تنتهي فيه كل محاولاته للهروب Home is not where you..."
وطن المرء ليس مكان ولادته و لكنه المكان الذي تنتهي فيه كل محاولاته للهروب
Home is not where you were born; home is where all your attempts to escape cease
- Naguib Mahfouz (1911-2006) - Egyptian writer & novelist
Quirky Interpretations of Everyday Objects by Vanessa McKeown


For the last few weeks, photographer and art director Vanessa McKeown has been sharing colorful, quirky interpretations of everyday objects on Instagram. McKeown imagines balloons as various fruits and vegetables and oranges are peeled to reveal unexpected objects. Clever visuals all around. You can also follow her on Tumblr.







4gifs: Horse brings his girlfriend a snack. [video]
GRK R4 Multi-Purpose Screws

I rebuilt my back stairs using pressurized treated lumber. While I was buying the wood at Home Depots I noticed a display of serious looking screws and since I know from experience that fastening wet pressurized 2x12s and 4x4s can be a challenge I thought I would try these. The main selling point was the “Zero Stripping” star drive (aka Torx) head and the “Easy Start” tip. My tool is a Milwaukee 18 volt screwdriver, which performed flawlessly. Though sometimes the screws were a little hard to start, overall they worked well. None of the boards split and there was no stripping. Another interesting feature is what they call “CEE Thread”. This allowed the screw to act like a lag screw. I used the #10 R4 Multi-Purpose Screws, both the 3 1/8” and the 2 ½”. If you have a project that does not require much finesse, these are the screws for you.
-- Dean Raffaelli
GRK R4 Multi-Purpose Screws
$12 – $15 per 100
Available from Amazon
Here's Part of the Reason Why These Male Lions Are Maneless
http://4erep-i-kosti.livejournal.com/4774700.html

Chemistry Matters
kirstendraws: Some of you may remember my popular bi, pan, and...







Some of you may remember my popular bi, pan, and asexuwhale trio from last year. Well, I’ve decided to redo the set and add EVEN MORE SEXUWHALES (and an aromanatee!)
Featuring:
Bisexuwhale - humpback whale
Homosexuwhale - sperm whale
Pansexuwhale - narwhal/narwhale
Polysexuwhale - beluga whale
Asexuwhale - killer whale/orca
Aromanatee - manateeAll are available in my Redbubble shop as stickers, shirts, mugs, and more; text-less versions will be added to my shop soon, too.